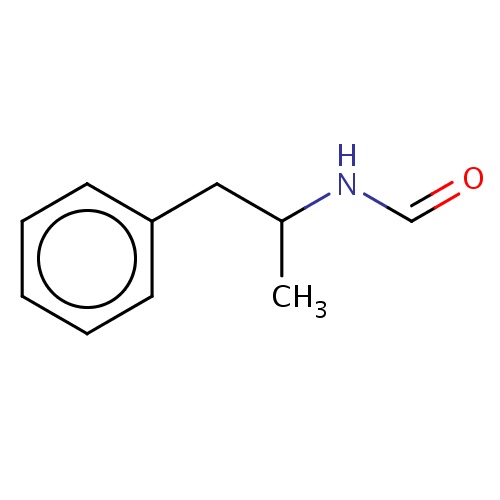
Common name
N-[(1R)-1-methyl-2-phenyl-ethyl]formamide
IUPAC name
N-[(1R)-1-methyl-2-phenyl-ethyl]formamide
SMILES
c1(ccccc1)CC(C)NC=O
Common name
N-[(1R)-1-methyl-2-phenyl-ethyl]formamide
IUPAC name
N-[(1R)-1-methyl-2-phenyl-ethyl]formamide
SMILES
c1(ccccc1)CC(C)NC=O
INCHI
InChI=1S/C10H13NO/c1-9(11-8-12)7-10-5-3-2-4-6-10/h2-6,8-9H,7H2,1H3,(H,11,12)/t9-/m1/s1
FORMULA
C10H13NO

Common name
N-[(1R)-1-methyl-2-phenyl-ethyl]formamide
IUPAC name
N-[(1R)-1-methyl-2-phenyl-ethyl]formamide
Molecular weight
163.216
clogP
2.043
clogS
-2.471
Frequency
0.0003
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
29.1
Number of Rings
1
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00376 | Ritonavir |

|
Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; Combined Inducers of CYP3A4 and P-glycoprotein; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Indicated in combination with other antiretroviral agents for the treatment of HIV-infection. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4gid_ligand_4_875.mol2 | 4gid | 1 | -7.60 | C[C@H](Cc1ccccc1)NC=O | 12 |
| 1j1a_ligand_4_1295.mol2 | 1j1a | 1 | -7.01 | C([C@H](C)NC=O)c1ccccc1 | 12 |
| 4u7q_ligand_4_1508.mol2 | 4u7q | 1 | -6.72 | C(=O)N[C@H](C)Cc1ccccc1 | 12 |
| 2o4s_ligand_4_1035.mol2 | 2o4s | 1 | -6.68 | C[C@@H](NC=O)Cc1ccccc1 | 12 |
| 1mui_ligand_4_1035.mol2 | 1mui | 1 | -6.62 | C[C@H](Cc1ccccc1)NC=O | 12 |
| 2q5k_ligand_4_1035.mol2 | 2q5k | 1 | -6.60 | C[C@H](Cc1ccccc1)NC=O | 12 |
| 2q54_ligand_4_870.mol2 | 2q54 | 1 | -6.59 | [C@H](Cc1ccccc1)(C)NC=O | 12 |
335 ,
34

