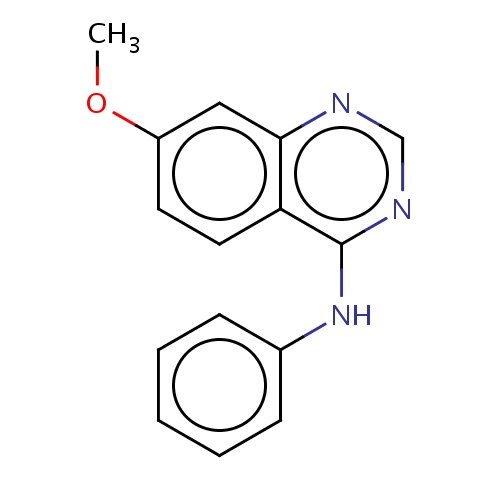
Common name
7-methoxy-N-phenyl-quinazolin-4-amine
IUPAC name
7-methoxy-N-phenyl-quinazolin-4-amine
SMILES
COc1ccc2c(c1)ncnc2Nc3ccccc3
Common name
7-methoxy-N-phenyl-quinazolin-4-amine
IUPAC name
7-methoxy-N-phenyl-quinazolin-4-amine
SMILES
COc1ccc2c(c1)ncnc2Nc3ccccc3
INCHI
InChI=1S/C15H13N3O/c1-19-12-7-8-13-14(9-12)16-10-17-15(13)18-11-5-3-2-4-6-11/h2-10H,1H3,(H,16,17,18)
FORMULA
C15H13N3O

Common name
7-methoxy-N-phenyl-quinazolin-4-amine
IUPAC name
7-methoxy-N-phenyl-quinazolin-4-amine
Molecular weight
251.283
clogP
2.674
clogS
-4.667
Frequency
0.0003
HBond Acceptor
3
HBond Donor
1
Total PolarSurface Area
47.04
Number of Rings
3
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00399 | Erlotinib |
|
Antineoplastic Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of patients with locally advanced or metastatic non-small cell lung cancer after failure of at least one prior chemotherapy regimen. Also for use, in combination with gemcitabine, as the first-line treatment of patients with locally advanced, unresectable or metastatic pancreatic cancer. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2vrx_ligand_4_179.mol2 | 2vrx | 1 | -8.26 | c1(ccccc1)Nc1c2c(cc(OC)cc2)ncn1 | 19 |
| 1di9_ligand_3_1.mol2 | 1di9 | 1 | -7.69 | O(C)c1ccc2c(ncnc2Nc2ccccc2)c1 | 19 |
| 2vrx_ligand_3_104.mol2 | 2vrx | 0.917808 | -8.26 | c1(ccccc1)Nc1c2c(cc(O)cc2)ncn1 | 18 |
| 4knx_ligand_3_19.mol2 | 4knx | 0.917808 | -7.44 | c1cc2c(cc1O)ncnc2Nc1ccccc1 | 18 |
| 4knr_ligand_2_11.mol2 | 4knr | 0.917808 | -7.43 | c1(ccccc1)Nc1c2ccc(cc2ncn1)O | 18 |
| 4e1k_ligand_2_4.mol2 | 4e1k | 0.917808 | -7.30 | c1cc2c(cc1O)ncnc2Nc1ccccc1 | 18 |
| 1kz8_ligand_3_38.mol2 | 1kz8 | 0.917808 | -6.45 | c1cc(ccc1)Nc1ncnc2c1ccc(O)c2 | 18 |
| 4npv_ligand_3_1.mol2 | 4npv | 0.890411 | -6.86 | CNc1c2c(cc(cc2)OC)ncn1 | 14 |
| 2vrx_ligand_3_110.mol2 | 2vrx | 0.871795 | -8.19 | c1(ccccc1)Nc1c2c(ccc(OC)c2)ncn1 | 19 |
| 1di9_ligand_3_3.mol2 | 1di9 | 0.871795 | -7.70 | O(C)c1cc2c(ncnc2Nc2ccccc2)cc1 | 19 |
107 ,
11

