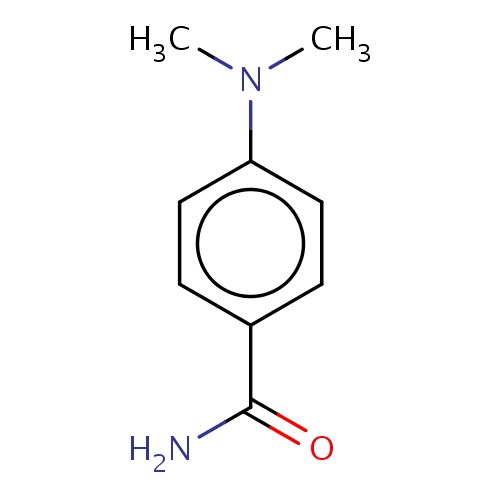
Common name
4-(dimethylamino)benzamide
IUPAC name
4-(dimethylamino)benzamide
SMILES
CN(c1ccc(cc1)C(=O)N)C
Common name
4-(dimethylamino)benzamide
IUPAC name
4-(dimethylamino)benzamide
SMILES
CN(c1ccc(cc1)C(=O)N)C
INCHI
InChI=1S/C9H12N2O/c1-11(2)8-5-3-7(4-6-8)9(10)12/h3-6H,1-2H3,(H2,10,12)
FORMULA
C9H12N2O

Common name
4-(dimethylamino)benzamide
IUPAC name
4-(dimethylamino)benzamide
Molecular weight
164.204
clogP
0.545
clogS
-1.483
Frequency
0.0003
HBond Acceptor
2
HBond Donor
2
Total PolarSurface Area
46.33
Number of Rings
1
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
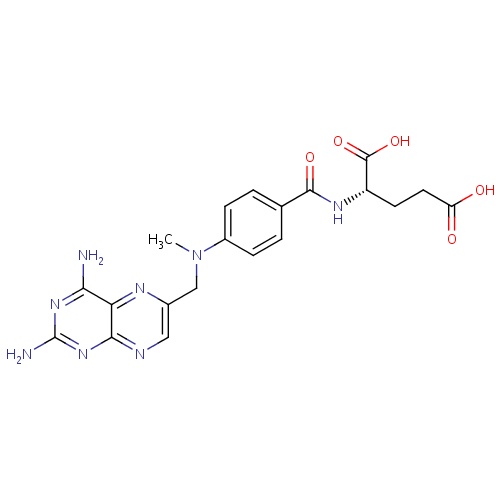
| FDBD00430 | Methotrexate |
|
Antineoplastic Agents; Antirheumatic Agents; Dermatologic Agents; Immunosuppressive Agents; Enzyme Inhibitors; Antimetabolites; Folic Acid Antagonists; Nucleic Acid Synthesis Inhibitors; Antimetabolites, Antineoplastic; Abortifacient Agents, Nonsteroidal; Abortifacient Agents; Antineoplastic and Immunomodulating Agents; Folic Acid Analogues; | Methotrexate is indicated in the treatment of gestational choriocarcinoma, chorioadenoma destruens and hydatidiform mole. In acute lymphocytic leukemia, methotrexate is indicated in the prophylaxis of meningeal leukemia and is used in maintenance therapy in combination with other chemotherapeutic agents. Methotrexate is also indicated in the treatment of meningeal leukemia. Methotrexate is used alone or in combination with other anticancer agents in the treatment of breast cancer, epidermoid cancers of the head and neck, advanced mycosis fungoides (cutaneous T cell lymphoma), and lung cancer, particularly squamous cell and small cell types. Methotrexate is also used in combination with other chemotherapeutic agents in the treatment of advanced stage non-Hodgkin's lymphomas. Methotrexate is indicated in the symptomatic control of severe, recalcitrant, disabling psoriasis. Methotrexate is indicated in the management of selected adults with severe, active rheumatoid arthritis (ACR criteria), or children with active polyarticular-course juvenile rheumatoid arthritis. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4kmz_ligand_3_15.mol2 | 4kmz | 1 | -7.06 | C(=O)(N)c1ccc(NC)cc1 | 11 |
| 4kn0_ligand_3_15.mol2 | 4kn0 | 1 | -7.06 | CN(c1ccc(cc1)C(=O)N)C | 12 |
| 4lrh_ligand_3_15.mol2 | 4lrh | 1 | -7.02 | N(c1ccc(cc1)C(=O)N)C | 11 |
| 4kn1_ligand_3_11.mol2 | 4kn1 | 1 | -6.96 | C(=O)(N)c1ccc(NC)cc1 | 11 |
| 4kn0_ligand_2_11.mol2 | 4kn0 | 1 | -6.95 | c1(ccc(cc1)NC)C(=O)N | 11 |
| 4u0m_ligand_3_130.mol2 | 4u0m | 1 | -6.83 | NC(=O)c1ccc(cc1)NC | 11 |
| 2oym_ligand_2_0.mol2 | 2oym | 1 | -6.82 | N(C)(C)c1ccc(cc1)C(=O)N | 12 |
| 3eig_ligand_3_15.mol2 | 3eig | 1 | -6.82 | CN(C)c1ccc(cc1)C(=O)N | 12 |
| 1dfo_ligand_3_15.mol2 | 1dfo | 1 | -6.79 | N(c1ccc(cc1)C(=O)N)C | 11 |
124 ,
13

