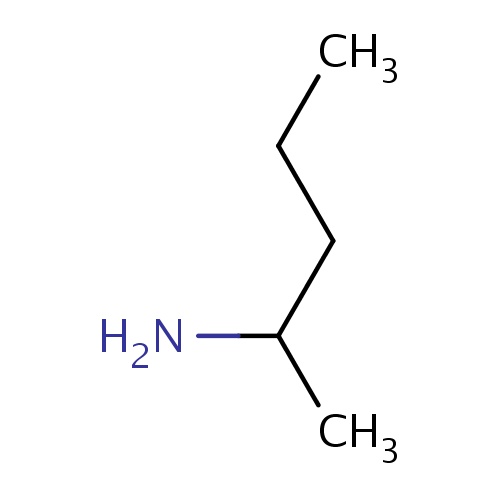
Common name
(2S)-pentan-2-amine
IUPAC name
(2S)-pentan-2-amine
SMILES
C(C)CC(C)N
Common name
(2S)-pentan-2-amine
IUPAC name
(2S)-pentan-2-amine
SMILES
C(C)CC(C)N
INCHI
InChI=1S/C5H13N/c1-3-4-5(2)6/h5H,3-4,6H2,1-2H3/t5-/m0/s1
FORMULA
C5H13N

Common name
(2S)-pentan-2-amine
IUPAC name
(2S)-pentan-2-amine
Molecular weight
87.163
clogP
0.443
clogS
-1.050
Frequency
0.0007
HBond Acceptor
0
HBond Donor
2
Total PolarSurface Area
26.02
Number of Rings
0
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
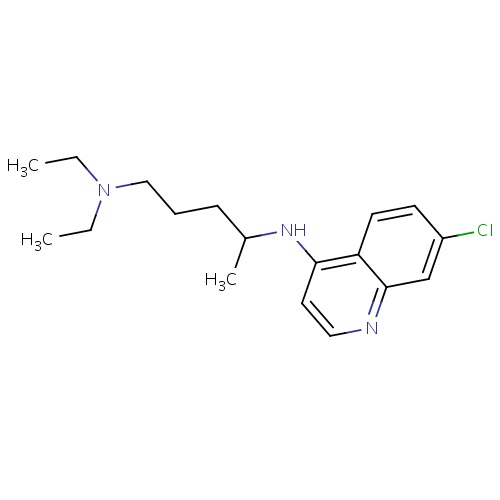
| FDBD00473 | Chloroquine |
|
Antirheumatic Agents; Antimalarials; Antiprotozoal Agents; Amebicides; Antiparasitic Products, Insecticides and Repellents; Aminoquinolines; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the suppressive treatment and for acute attacks of malaria due to P. vivax, P.malariae, P. ovale, and susceptible strains of P. falciparum, Second-line agent in treatment of Rheumatoid Arthritis. |
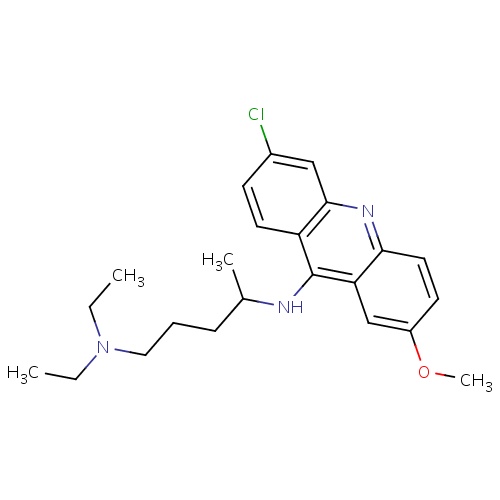
| FDBD00953 | Quinacrine |
|
Antineoplastic Agents; Enzyme Inhibitors; Antimalarials; Antiprotozoal Agents; Antinematodal Agents; Anthelmintics; Anticestodal Agents; Antiparasitic Products, Insecticides and Repellents; Agents Against Protozoal Diseases; CYP3A4 Inhibitors; | For the treatment of giardiasis and cutaneous leishmaniasis and the management of malignant effusions. |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2fdp_ligand_3_88.mol2 | 2fdp | 1 | -6.26 | C[C@H](CCC)[NH3+] | 6 |
| 1jqe_ligand_4_91.mol2 | 1jqe | 1 | -6.08 | CCC[C@@H](C)[NH3+] | 6 |
| 1cet_ligand_4_35.mol2 | 1cet | 1 | -5.45 | CCC[C@@H](C)[NH3+] | 6 |
| 2v0z_ligand_4_3381.mol2 | 2v0z | 0.888889 | -6.59 | C(C[NH3+])[C@@H](C(C)C)C | 8 |
| 2v12_ligand_4_2730.mol2 | 2v12 | 0.888889 | -6.48 | C(C)(C)[C@H](CC[NH3+])C | 8 |
395 ,
40

