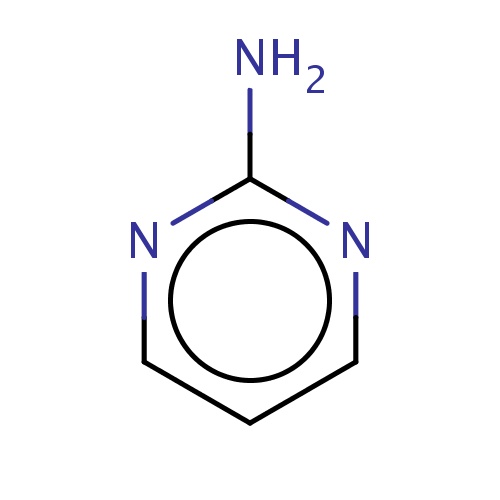
Common name
pyrimidin-2-amine
IUPAC name
pyrimidin-2-amine
SMILES
Nc1ncccn1
Common name
pyrimidin-2-amine
IUPAC name
pyrimidin-2-amine
SMILES
Nc1ncccn1
INCHI
InChI=1S/C4H5N3/c5-4-6-2-1-3-7-4/h1-3H,(H2,5,6,7)
FORMULA
C4H5N3

Common name
pyrimidin-2-amine
IUPAC name
pyrimidin-2-amine
Molecular weight
95.103
clogP
0.377
clogS
-0.751
Frequency
0.0024
HBond Acceptor
2
HBond Donor
2
Total PolarSurface Area
51.8
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00484 | Imatinib |
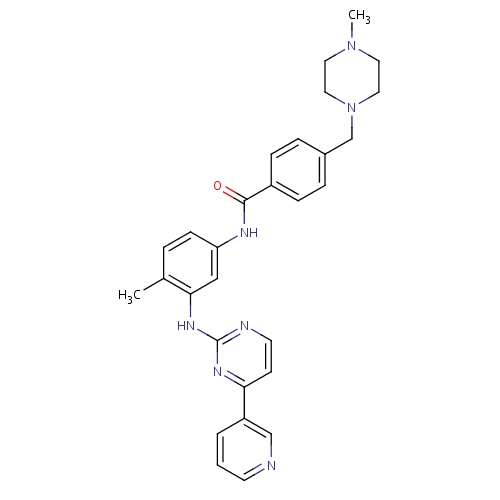
|
Antineoplastic Agents; Immunosuppressive Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the treatment of Philadelphia chromosome positive chronic myeloid leukemia (Ph+ CML), Ph+ acute lymphoblastic leukaemia, myelodysplastic/myeloproliferative diseases, aggressive systemic mastocytosis, hypereosinophilic syndrome and/or chronic eosinophilic leukemia (CEL), dermatofibrosarcoma protuberans, and malignant gastrointestinal stromal tumors (GIST). |
| FDBD01336 | Nilotinib |
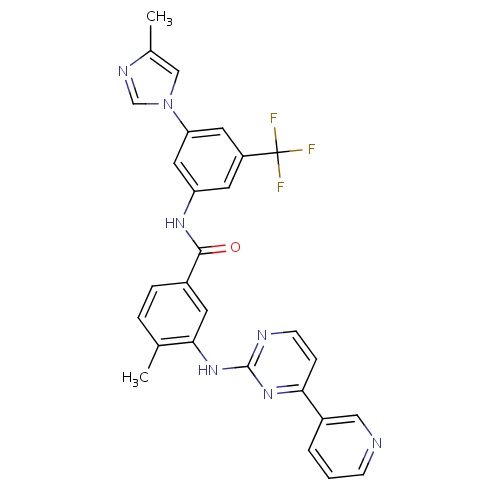
|
Antineoplastic Agents; Immunosuppressive Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the potential treatment of various leukemias, including chronic myeloid leukemia (CML). |
| FDBD01361 | Silver sulfadiazine |
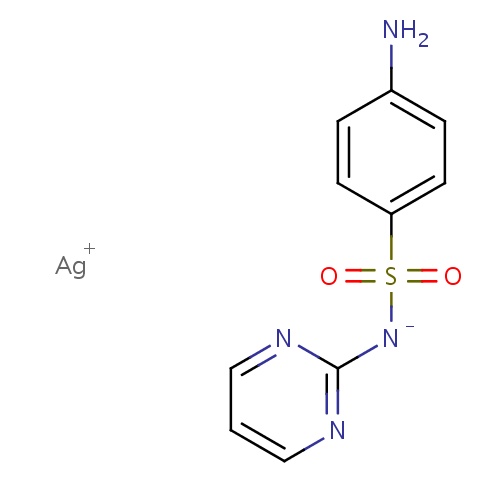
|
Anti-Bacterial Agents; Anti-Infective Agents, Local; Sulfonamides; Antibiotics, Topical; Dermatologicals; | Indicated as an adjunct for the prevention and treatment of wound sepsis in patients with second- and third-degree burns. |
| FDBD01419 | Pazopanib |

|
Antineoplastic Agents; Immunosuppressive Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | Treatment of advanced renal cell cancer and advanced soft tissue sarcoma (in patients previously treated with chemotherapy) . |
| FDBD01544 | Rilpivirine |

|
Anti-HIV Agents; Reverse Transcriptase Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Non-Nucleoside Reverse Transcriptase Inhibitors; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Treatment of HIV-1 infections in treatment-naive patients with HIV-1 RNA |
| FDBD01573 | Dabrafenib |

|
Antineoplastic Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; | Dabrafenib is indicated for the treatment of patients with unresectable or metastatic melanoma with BRAF V600E mutation as detected by an FDA-approved test. |
| FDBD01810 | Osimertinib |
|
Antineoplastic Agents; Protein Kinase Inhibitors; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP3A4 Inhibitors; | Osimertinib is indicated for the treatment of patients with metastatic epidermal growth factor receptor (EGFR) T790M mutation-positive non-small cell lung cancer (NSCLC), as detected by an FDA- approved test, who have progressed on or after EGFR-TKI therapy. |
7 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2w70_ligand_frag_1.mol2 | 2w70 | 1 | -6.33 | c1ccnc(n1)N | 7 |
| 4n1t_ligand_frag_0.mol2 | 4n1t | 1 | -6.28 | c1cnc(nc1)N | 7 |
| 2w71_ligand_frag_3.mol2 | 2w71 | 1 | -6.27 | c1ccnc(n1)N | 7 |
| 4qac_ligand_frag_1.mol2 | 4qac | 1 | -6.25 | c1ccnc(n1)N | 7 |
| 4qab_ligand_frag_1.mol2 | 4qab | 1 | -6.23 | c1ccnc(n1)N | 7 |
| 2c5y_ligand_1_3.mol2 | 2c5y | 1 | -6.20 | c1nc(ncc1)N | 7 |
| 1pxm_ligand_1_1.mol2 | 1pxm | 1 | -6.19 | c1(ncccn1)N | 7 |
| 4d0x_ligand_frag_3.mol2 | 4d0x | 1 | -6.19 | c1ccnc(n1)N | 7 |
178 ,
18

