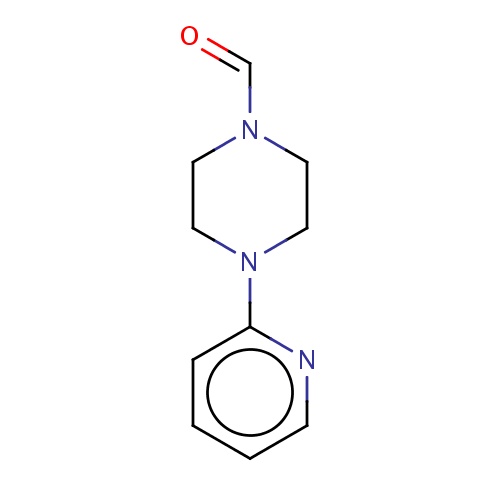
Common name
4-(2-pyridyl)piperazine-1-carbaldehyde
IUPAC name
4-(2-pyridyl)piperazine-1-carbaldehyde
SMILES
O=CN1CCN(CC1)c2ncccc2
Common name
4-(2-pyridyl)piperazine-1-carbaldehyde
IUPAC name
4-(2-pyridyl)piperazine-1-carbaldehyde
SMILES
O=CN1CCN(CC1)c2ncccc2
INCHI
InChI=1S/C10H13N3O/c14-9-12-5-7-13(8-6-12)10-3-1-2-4-11-10/h1-4,9H,5-8H2
FORMULA
C10H13N3O

Common name
4-(2-pyridyl)piperazine-1-carbaldehyde
IUPAC name
4-(2-pyridyl)piperazine-1-carbaldehyde
Molecular weight
191.230
clogP
0.683
clogS
-1.152
Frequency
0.0003
HBond Acceptor
3
HBond Donor
0
Total PolarSurface Area
36.44
Number of Rings
2
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00569 | Delavirdine |
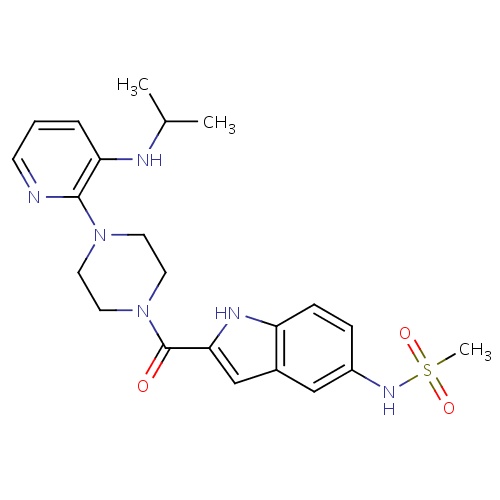
|
Anti-HIV Agents; Reverse Transcriptase Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Non-Nucleoside Reverse Transcriptase Inhibitors; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of HIV-1 infection in combination with appropriate antiretroviral agents when therapy is warranted. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1rd4_ligand_1_2.mol2 | 1rd4 | 0.982759 | -6.31 | c1ccnc(c1)N1CCN(CC1)C(=O)C | 15 |
| 4y2q_ligand_1_1.mol2 | 4y2q | 0.894737 | -7.18 | c1ccc(nc1)N1CC[NH2+]CC1 | 12 |
| 4bfz_ligand_2_20.mol2 | 4bfz | 0.894737 | -6.66 | [NH+]1(CCN(CC1)c1ncccc1)C | 13 |
| 4bfy_ligand_2_11.mol2 | 4bfy | 0.894737 | -6.65 | C[NH+]1CCN(c2ccccn2)CC1 | 13 |
| 4bfz_ligand_1_6.mol2 | 4bfz | 0.894737 | -6.55 | [NH2+]1CCN(CC1)c1ncccc1 | 12 |
| 4bfy_ligand_1_5.mol2 | 4bfy | 0.894737 | -6.54 | c1ccc(nc1)N1CC[NH2+]CC1 | 12 |
| 2ynd_ligand_1_1.mol2 | 2ynd | 0.894737 | -6.47 | C1CN(CC[NH2+]1)c1ccccn1 | 12 |
100 ,
11

