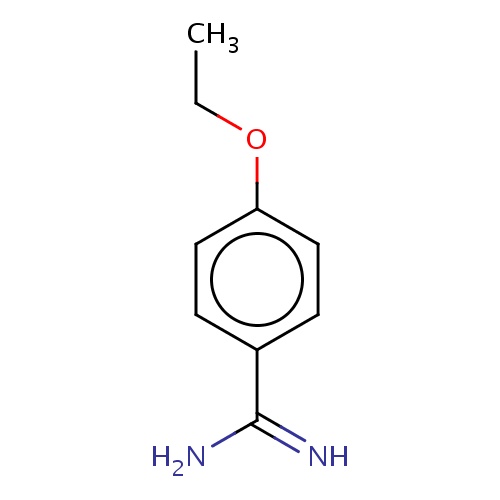
Common name
4-ethoxybenzamidine
IUPAC name
4-ethoxybenzamidine
SMILES
C(C)Oc1ccc(cc1)C(=N)N
Common name
4-ethoxybenzamidine
IUPAC name
4-ethoxybenzamidine
SMILES
C(C)Oc1ccc(cc1)C(=N)N
INCHI
InChI=1S/C9H12N2O/c1-2-12-8-5-3-7(4-6-8)9(10)11/h3-6H,2H2,1H3,(H3,10,11)
FORMULA
C9H12N2O

Common name
4-ethoxybenzamidine
IUPAC name
4-ethoxybenzamidine
Molecular weight
164.204
clogP
1.439
clogS
-2.049
Frequency
0.0007
HBond Acceptor
1
HBond Donor
3
Total PolarSurface Area
59.1
Number of Rings
1
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00602 | Pentamidine |
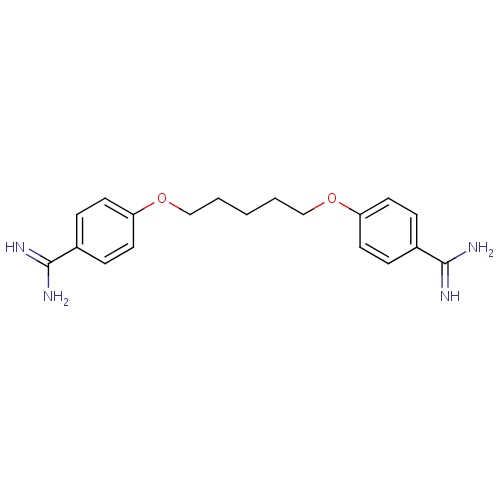
|
Antifungal Agents; Antiprotozoal Agents; Trypanocidal Agents; Antiparasitic Products, Insecticides and Repellents; Agents Against Protozoal Diseases; Agents Against Leishmaniasis and Trypanosomiasis; Aromatic Diamidines; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of pneumonia due to . |
| FDBD03262 | propamidine |

|
Fungicide | Fungicide |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3cr5_ligand_3_55.mol2 | 3cr5 | 1 | -6.68 | O(CC)c1ccc(cc1)C(=N)N | 12 |
| 3gy3_ligand_3_55.mol2 | 3gy3 | 1 | -6.37 | CCOc1ccc(cc1)C(=N)N | 12 |
| 4fqo_ligand_3_0.mol2 | 4fqo | 1 | -5.70 | C(C)Oc1ccc(C(=N)N)cc1 | 12 |
| 3cr5_ligand_2_26.mol2 | 3cr5 | 0.952381 | -6.56 | O(C)c1ccc(cc1)C(=N)N | 11 |
| 3gy3_ligand_2_26.mol2 | 3gy3 | 0.952381 | -6.19 | c1(ccc(cc1)C(=N)N)OC | 11 |
| 4fqo_ligand_2_0.mol2 | 4fqo | 0.952381 | -5.55 | COc1ccc(C(=N)N)cc1 | 11 |
| 4o9v_ligand_2_14.mol2 | 4o9v | 0.869565 | -6.50 | c1cccc(c1)Oc1ccc(cc1)C(=N)N | 16 |
227 ,
23

