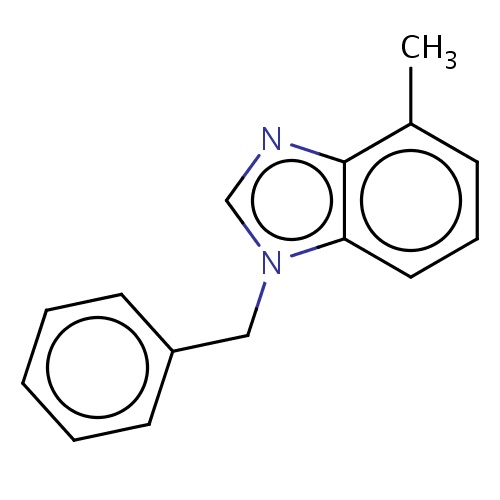
Common name
1-benzyl-4-methyl-benzimidazole
IUPAC name
1-benzyl-4-methyl-benzimidazole
SMILES
n1(c2c(nc1)c(ccc2)C)Cc3ccccc3
Common name
1-benzyl-4-methyl-benzimidazole
IUPAC name
1-benzyl-4-methyl-benzimidazole
SMILES
n1(c2c(nc1)c(ccc2)C)Cc3ccccc3
INCHI
InChI=1S/C15H14N2/c1-12-6-5-9-14-15(12)16-11-17(14)10-13-7-3-2-4-8-13/h2-9,11H,10H2,1H3
FORMULA
C15H14N2

Common name
1-benzyl-4-methyl-benzimidazole
IUPAC name
1-benzyl-4-methyl-benzimidazole
Molecular weight
223.293
clogP
2.215
clogS
-4.043
Frequency
0.0003
HBond Acceptor
0
HBond Donor
1
Total PolarSurface Area
19.67
Number of Rings
3
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00820 | Telmisartan |

|
Antihypertensive Agents; Angiotensin II Type 1 Receptor Blockers; Cardiovascular System; Angiotensin II Antagonists, Plain; Agents Acting on the Renin-Angiotensin System; Angiotensin II Antagonists and Diuretics; Angiotensin II Antagonists and Calcium Channel Blockers; Cytochrome P-450 CYP2C19 Inducers; Angiotensin II Receptor Antagonists; | Used alone or in combination with other classes of antihypertensives for the treatment of hypertension. Also used in the treatment of diabetic nephropathy in hypertensive patients with type 2 diabetes mellitus, as well as the treatment of congestive heart failure (only in patients who cannot tolerate ACE inhibitors). |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4jvb_ligand_2_0.mol2 | 4jvb | 0.859155 | -8.21 | [n+]1(c2ccccc2[nH]c1)Cc1ccccc1 | 16 |
| 4jv6_ligand_2_0.mol2 | 4jv6 | 0.859155 | -8.19 | [n+]1(c2ccccc2[nH]c1)Cc1ccccc1 | 16 |
| 5ea3_ligand_1_1.mol2 | 5ea3 | 0.760563 | -5.99 | c1(cccc2c1[nH]c[n+]2C)C | 11 |
| 1g2m_ligand_2_9.mol2 | 1g2m | 0.730337 | -7.91 | [nH]1c[n+](c2c1cc(cc2)C[n+]1c([nH]c2c1cccc2)C)C | 21 |
| 1g32_ligand_2_9.mol2 | 1g32 | 0.730337 | -7.75 | [nH]1c2c([n+](c1)C)ccc(c2)C[n+]1c([nH]c2c1cccc2)C | 21 |
| 1g36_ligand_2_9.mol2 | 1g36 | 0.730337 | -6.91 | [n+]1(c([nH]c2c1cccc2)C)Cc1ccc2c([nH]c[n+]2C)c1 | 21 |
| 1g2m_ligand_3_9.mol2 | 1g2m | 0.722222 | -8.14 | [nH]1c([n+](c2c1cc(cc2)C[n+]1c([nH]c2c1cccc2)C)C)C | 22 |
101 ,
11

