
Common name
7H-purine
IUPAC name
7H-purine
SMILES
[nH]1c2cncnc2nc1
Common name
7H-purine
IUPAC name
7H-purine
SMILES
[nH]1c2cncnc2nc1
INCHI
InChI=1S/C5H4N4/c1-4-5(8-2-6-1)9-3-7-4/h1-3H,(H,6,7,8,9)
FORMULA
C5H4N4

Common name
7H-purine
IUPAC name
7H-purine
Molecular weight
120.112
clogP
1.231
clogS
-1.663
Frequency
0.0010
HBond Acceptor
3
HBond Donor
1
Total PolarSurface Area
54.46
Number of Rings
2
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00847 | Azathioprine |

|
Antirheumatic Agents; Immunosuppressive Agents; Antimetabolites; Antimetabolites, Antineoplastic; Antineoplastic and Immunomodulating Agents; | For use in rheumatoid arthritis, preventing renal transplant rejection, Crohn's disease, and colitis. |
| FDBD01417 | Cangrelor |
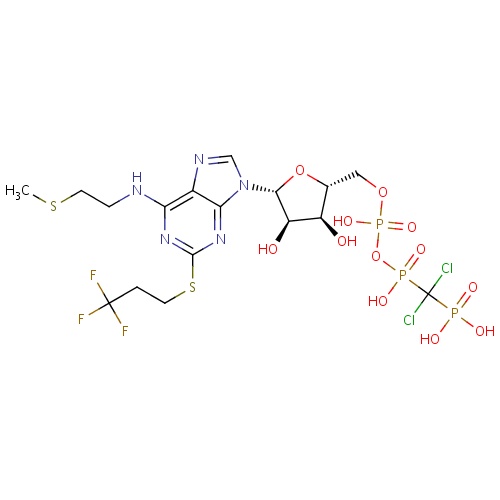
|
Platelet Aggregation Inhibitors; Antithrombotic Agents; Blood and Blood Forming Organs; Platelet Aggregation Inhibitors Excl. Heparin; Purinergic P2Y Receptor Antagonists; | For use as an adjunct to percutaneous coronary intervention (PCI) for reducing the risk of periprocedural myocardial infarction (MI), repeat coronary revascularization, and stent thrombosis (ST) in patients in who have not been treated with a P2Y12 platelet inhibitor and are not being given a glycoprotein IIb/IIIa inhibitor. |
| FDBD01649 | Idelalisib |
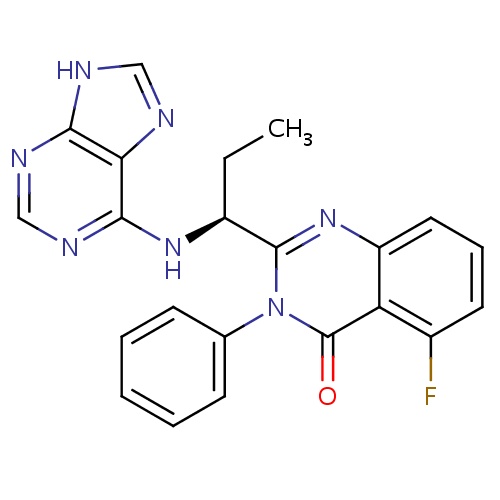
|
Antineoplastic Agents; Immunosuppressive Agents; Enzyme Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP3A4 Inhibitors; | Idelalisib is indicated in the treatment of chronic lymphocytic leukemia (CLL), relapsed follicular B-cell non-Hodgkin lymphoma (FL), and relapsed small lymphocytic lymphoma (SLL). For the treatment of relapsed CLL, it is currently indicated as a second-line agent in combination with rituximab in patients for whom rituximab alone would be considered appropriate therapy due to other co-morbidities, while in the treatment of FL and SLL it is intended to be used in patients who have received at least two prior systemic therapies. |
3 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2uw0_ligand_frag_3.mol2 | 2uw0 | 1 | -6.35 | c1ncnc2[nH+]c[nH]c12 | 9 |
| 3hvk_ligand_frag_0.mol2 | 3hvk | 1 | -6.35 | [nH+]1c2c([nH]c1)cncn2 | 9 |
| 3hvh_ligand_frag_6.mol2 | 3hvh | 1 | -6.30 | [nH+]1c[nH]c2c1ncnc2 | 9 |
| 3hvi_ligand_frag_6.mol2 | 3hvi | 1 | -6.30 | [nH+]1c[nH]c2c1ncnc2 | 9 |
| 3hvj_ligand_frag_6.mol2 | 3hvj | 1 | -6.29 | [nH+]1c[nH]c2c1ncnc2 | 9 |
109 ,
11

