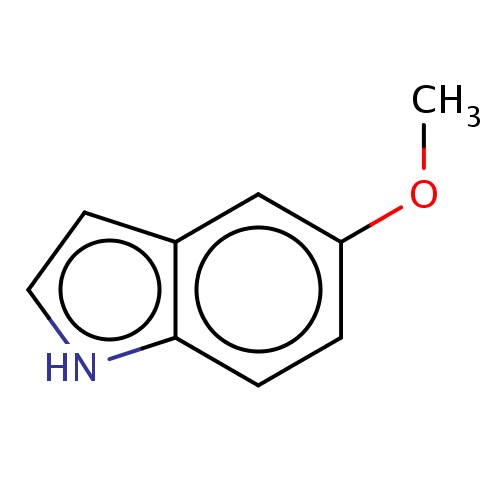
Common name
5-methoxy-1H-indole
IUPAC name
5-methoxy-1H-indole
SMILES
[nH]1c2c(cc1)cc(cc2)OC
Common name
5-methoxy-1H-indole
IUPAC name
5-methoxy-1H-indole
SMILES
[nH]1c2c(cc1)cc(cc2)OC
INCHI
InChI=1S/C9H9NO/c1-11-8-2-3-9-7(6-8)4-5-10-9/h2-6,10H,1H3
FORMULA
C9H9NO

Common name
5-methoxy-1H-indole
IUPAC name
5-methoxy-1H-indole
Molecular weight
147.174
clogP
2.531
clogS
-2.633
Frequency
0.0003
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
25.02
Number of Rings
2
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
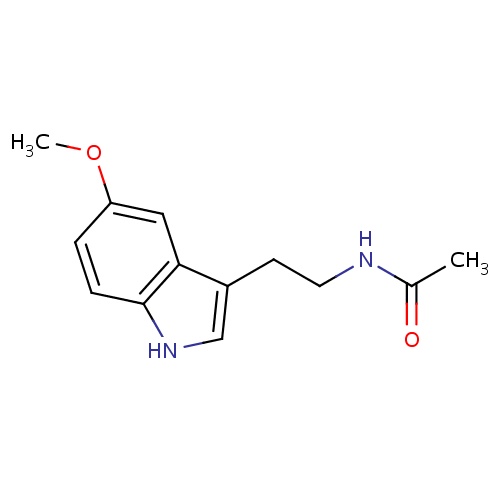
| FDBD00916 | Melatonin |
|
Antioxidants; Hypnotics and Sedatives; Central Nervous System Depressants; Nervous System; Psycholeptics; Melatonin Receptor Agonists; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; | Used orally for jet lag, insomnia, shift-work disorder, circadian rhythm disorders in the blind (evidence for efficacy), and benzodiazepine and nicotine withdrawal. Evidence indicates that melatonin is likely effective for treating circadian rhythm sleep disorders in blind children and adults. It has received FDA orphan drug status as an oral medication for this use. A number of studies have shown that melatonin may be effective for treating sleep-wake cycle disturbances in children and adolescents with mental retardation, autism, and other central nervous system disorders. It appears to decrease the time to fall asleep in children with developmental disabilities, such as cerebral palsy, autism, and mental retardation. It may also improve secondary insomnia associated with various sleep-wake cycle disturbances. Other possible uses for which there is some evidence for include: benzodiazepine withdrawal, cluster headache, delayed sleep phase syndrome (DSPS), primary insomnia, jet lag, nicotine withdrawal, preoperative anxiety and sedation, prostate cancer, solid tumors (when combined with IL-2 therapy in certain cancers), sunburn prevention (topical use), tardive dyskinesia, thrombocytopenia associated with cancer, chemotherapy and other disorders. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1db5_ligand_4_2.mol2 | 1db5 | 1 | -7.40 | [C@H]1(CC[C@@H]2[C@H](C1)[C@H]([C@@H]([N@H+]2C)C)C)OC | 14 |
| 3fuj_ligand_2_5.mol2 | 3fuj | 1 | -7.40 | [C@@H]1(CC[C@@H]2[C@@H](C1)CC[NH2+]2)OC | 11 |
| 1db4_ligand_4_2.mol2 | 1db4 | 1 | -7.39 | C[C@H]1[C@@H]2[C@@H](CC[C@@H](C2)OC)[N@H+]([C@H]1C)C | 14 |
| 3fuk_ligand_2_5.mol2 | 3fuk | 1 | -7.32 | [C@@H]1(CC[C@H]2[C@H](C1)CC[NH2+]2)OC | 11 |
| 1db5_ligand_3_8.mol2 | 1db5 | 1 | -7.28 | [C@H]1(CC[C@@H]2[C@H](C1)[C@H]([C@@H]([NH2+]2)C)C)OC | 13 |
| 1db4_ligand_3_9.mol2 | 1db4 | 1 | -7.27 | C[C@H]1[C@@H]2[C@@H](CC[C@@H](C2)OC)[NH2+][C@H]1C | 13 |
121 ,
13

