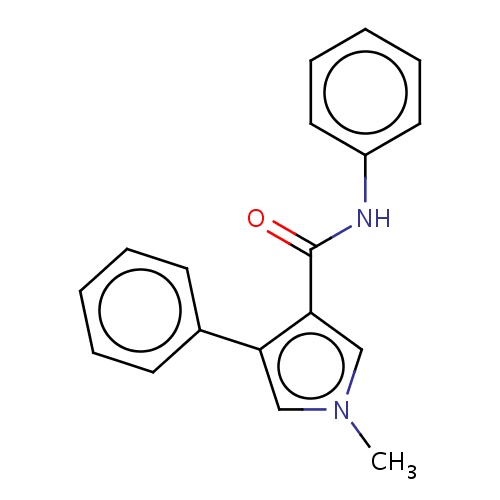
Common name
1-methyl-N,4-diphenyl-pyrrole-3-carboxamide
IUPAC name
1-methyl-N,4-diphenyl-pyrrole-3-carboxamide
SMILES
c1(ccccc1)NC(=O)c2cn(cc2c3ccccc3)C
Common name
1-methyl-N,4-diphenyl-pyrrole-3-carboxamide
IUPAC name
1-methyl-N,4-diphenyl-pyrrole-3-carboxamide
SMILES
c1(ccccc1)NC(=O)c2cn(cc2c3ccccc3)C
INCHI
InChI=1S/C18H16N2O/c1-20-12-16(14-8-4-2-5-9-14)17(13-20)18(21)19-15-10-6-3-7-11-15/h2-13H,1H3,(H,19,21)
FORMULA
C18H16N2O

Common name
1-methyl-N,4-diphenyl-pyrrole-3-carboxamide
IUPAC name
1-methyl-N,4-diphenyl-pyrrole-3-carboxamide
Molecular weight
281.372
clogP
2.727
clogS
-4.488
Frequency
0.0003
HBond Acceptor
1
HBond Donor
2
Total PolarSurface Area
33.54
Number of Rings
3
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00927 | Atorvastatin |
|
Anticholesteremic Agents; Hydroxymethylglutaryl-CoA Reductase Inhibitors; Dipeptidyl-Peptidase IV Inhibitors; HMG CoA Reductase Inhibitors; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3cct_ligand_3_35.mol2 | 3cct | 0.886364 | -7.15 | c1cccc(c1)[C@H]1C[N@@H+](C)C[C@@H]1C(=O)Nc1ccccc1O | 22 |
| 3cct_ligand_2_7.mol2 | 3cct | 0.74359 | -6.50 | c1cccc(c1)[C@H]1C[N@@H+](C)C[C@@H]1C(=O)N | 15 |
| 3cct_ligand_3_19.mol2 | 3cct | 0.734177 | -6.74 | c1cccc(c1)[C@H]1C[N@@H+](CC)C[C@@H]1C(=O)N | 16 |
| 4lwu_ligand_frag_0.mol2 | 4lwu | 0.684685 | -7.31 | N1c2cc(Cl)ccc2[C@@]2(C1=O)C[NH2+]CC2 | 15 |
| 4lwu_ligand_1_0.mol2 | 4lwu | 0.666667 | -7.57 | N1c2cc(Cl)ccc2[C@@]2(C1=O)[C@@H]([NH2+]CC2)C | 16 |
| 3cct_ligand_3_13.mol2 | 3cct | 0.659091 | -7.30 | c1cccc(c1)[C@H]1C[N@@H+](C)[C@@H](C(C)C)[C@@H]1C(=O)N | 18 |
103 ,
11

