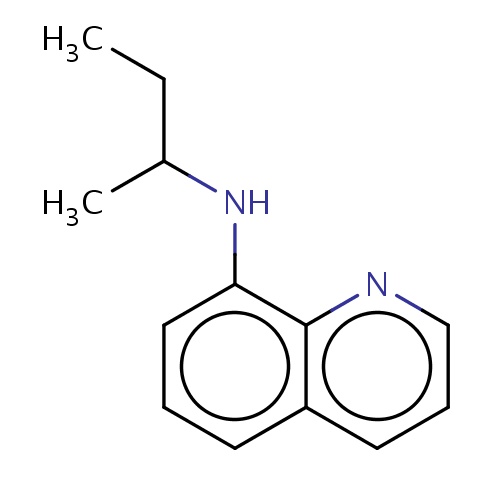
Common name
N-[(1R)-1-methylpropyl]quinolin-8-amine
IUPAC name
N-[(1R)-1-methylpropyl]quinolin-8-amine
SMILES
CCC(C)Nc1c2ncccc2ccc1
Common name
N-[(1R)-1-methylpropyl]quinolin-8-amine
IUPAC name
N-[(1R)-1-methylpropyl]quinolin-8-amine
SMILES
CCC(C)Nc1c2ncccc2ccc1
INCHI
InChI=1S/C13H16N2/c1-3-10(2)15-12-8-4-6-11-7-5-9-14-13(11)12/h4-10,15H,3H2,1-2H3/t10-/m1/s1
FORMULA
C13H16N2

Common name
N-[(1R)-1-methylpropyl]quinolin-8-amine
IUPAC name
N-[(1R)-1-methylpropyl]quinolin-8-amine
Molecular weight
200.280
clogP
2.985
clogS
-4.076
Frequency
0.0003
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
24.92
Number of Rings
2
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00937 | Primaquine |
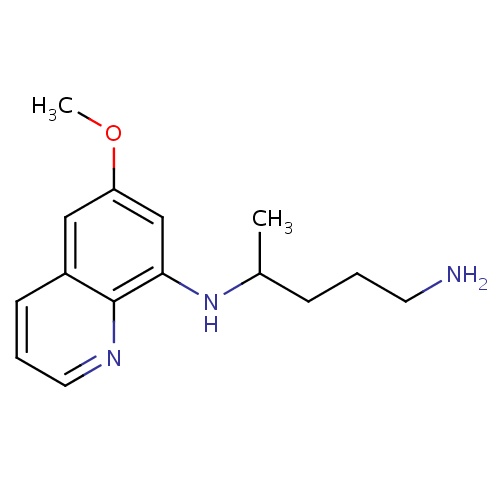
|
Antimalarials; Antiprotozoal Agents; Antiparasitic Products, Insecticides and Repellents; Aminoquinolines; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of malaria. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4mib_ligand_1_3.mol2 | 4mib | 0.875 | -7.18 | c1cccc2c1ncc(c2)N1CCCC1 | 15 |
| 4mib_ligand_2_12.mol2 | 4mib | 0.859649 | -7.40 | c1cccc2c1ncc(c2)N1CC[C@@H](C1)C | 16 |
| 4yrs_ligand.mol2 | 4yrs | 0.774194 | -6.95 | n1c2ccccc2cc(c1)NC(=O)CC | 16 |
| 4cqg_ligand_2_9.mol2 | 4cqg | 0.705882 | -7.72 | c1(ccnc2c1nccc2)NC1CCCCC1 | 17 |
| 4cqg_ligand_3_10.mol2 | 4cqg | 0.695652 | -7.94 | c1(ccnc2c1nccc2)N[C@@H]1CC[C@H](CC1)C | 18 |
| 4yrr_ligand.mol2 | 4yrr | 0.688525 | -6.95 | O=C(C)Nc1cc2c(nc1)cccc2 | 15 |
| 4mib_ligand_2_9.mol2 | 4mib | 0.680556 | -7.18 | O(C)c1cccc2c1cc(cn2)N1CCCC1 | 17 |
| 4mib_ligand_3_16.mol2 | 4mib | 0.671233 | -7.40 | O(C)c1cccc2c1cc(cn2)N1CC[C@@H](C1)C | 18 |
| 4gg5_ligand_1_0.mol2 | 4gg5 | 0.666667 | -7.58 | c1(cc2c(nc1)cccc2)N1CC[NH+](CC1)C | 17 |
| 4tpp_ligand_1_3.mol2 | 4tpp | 0.655738 | -7.69 | C1CN(C1)c1ccc2ccccc2n1 | 14 |
103 ,
11

