Common name
(1R,2R,3aS,5E,6aS)-5-ethylidene-1-[(E,3S)-3-hydroxypent-1-enyl]-2,3,3a,4,6,6a-hexahydro-1H-pentalen-2-ol
IUPAC name
(1R,2R,3aS,5E,6aS)-5-ethylidene-1-[(E,3S)-3-hydroxypent-1-enyl]-2,3,3a,4,6,6a-hexahydro-1H-pentalen-2-ol
SMILES
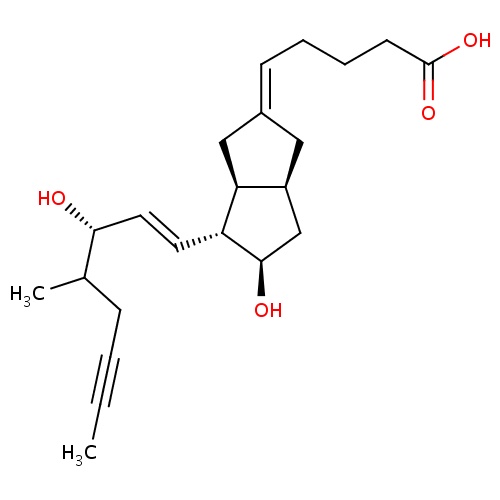
C(C)C(O)C=CC1C2C(CC1O)CC(=CC)C2
Common name
(1R,2R,3aS,5E,6aS)-5-ethylidene-1-[(E,3S)-3-hydroxypent-1-enyl]-2,3,3a,4,6,6a-hexahydro-1H-pentalen-2-ol
IUPAC name
(1R,2R,3aS,5E,6aS)-5-ethylidene-1-[(E,3S)-3-hydroxypent-1-enyl]-2,3,3a,4,6,6a-hexahydro-1H-pentalen-2-ol
SMILES
C(C)C(O)C=CC1C2C(CC1O)CC(=CC)C2
INCHI
InChI=1S/C15H24O2/c1-3-10-7-11-9-15(17)13(14(11)8-10)6-5-12(16)4-2/h3,5-6,11-17H,4,7-9H2,1-2H3/b6-5+,10-3+/t11-,12-,13+,14-,15+/m0/s1
FORMULA
C15H24O2

Common name
(1R,2R,3aS,5E,6aS)-5-ethylidene-1-[(E,3S)-3-hydroxypent-1-enyl]-2,3,3a,4,6,6a-hexahydro-1H-pentalen-2-ol
IUPAC name
(1R,2R,3aS,5E,6aS)-5-ethylidene-1-[(E,3S)-3-hydroxypent-1-enyl]-2,3,3a,4,6,6a-hexahydro-1H-pentalen-2-ol
Molecular weight
236.350
clogP
2.653
clogS
-1.381
Frequency
0.0003
HBond Acceptor
2
HBond Donor
2
Total PolarSurface Area
40.46
Number of Rings
2
Rotatable Bond
3
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3e6y_ligand_1_6.mol2 | 3e6y | 0.789474 | -7.86 | C(C)(C)[C@H]1[C@@H]2C[C@@H]([C@@H]([C@H]3C(=C[C@]2(CC1)C)[C@@H](CC3)O)C)O | 21 |
| 3e6y_ligand_1_7.mol2 | 3e6y | 0.789474 | -7.86 | C[C@@]1(C2=C[C@@]3([C@@H](C[C@@H]([C@@H]([C@@H]2CC1)C)O)CCC3)C)O | 19 |
| 3e6y_ligand_frag_6.mol2 | 3e6y | 0.789474 | -7.50 | C1[C@H]2CCC[C@@]2(C=C2[C@@H](CC[C@H]2[C@H]([C@H]1O)C)O)C | 18 |
| 1o9e_ligand_2_1.mol2 | 1o9e | 0.763158 | -8.00 | C[C@@H]1C2=C[C@@]3([C@@H]([C@@H](C[C@@H]3O)CC)C[C@@H]([C@@H]([C@@H]2CC1)C)O)C | 21 |
| 1o9e_ligand_1_2.mol2 | 1o9e | 0.763158 | -7.76 | C[C@@H]1C2=C[C@@]3([C@@H](CC[C@@H]3O)C[C@@H]([C@@H]([C@@H]2CC1)C)O)C | 19 |
| 1o9e_ligand_1_0.mol2 | 1o9e | 0.763158 | -7.69 | [C@@H]12[C@@H](C[C@@H]([C@@]1(C=C1CCC[C@H]1[C@H]([C@H](C2)O)C)C)O)CC | 20 |
| 1o9e_ligand_frag_0.mol2 | 1o9e | 0.763158 | -7.45 | [C@@H]12CC[C@@H]([C@@]1(C=C1CCC[C@H]1[C@H]([C@H](C2)O)C)C)O | 18 |
| 2am9_ligand.mol2 | 2am9 | 0.75 | -10.14 | C1C[C@@H](O)C=C2CC[C@@H]3[C@@H]([C@@]12C)CC[C@]1([C@H]3CC[C@@H]1O)C | 22 |