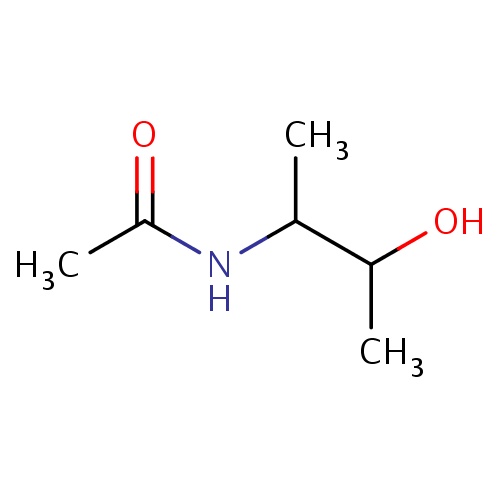
Common name
N-[(1R,2R)-2-hydroxy-1-methyl-propyl]acetamide
IUPAC name
N-[(1R,2R)-2-hydroxy-1-methyl-propyl]acetamide
SMILES
CC(=O)NC(C(O)C)C
Common name
N-[(1R,2R)-2-hydroxy-1-methyl-propyl]acetamide
IUPAC name
N-[(1R,2R)-2-hydroxy-1-methyl-propyl]acetamide
SMILES
CC(=O)NC(C(O)C)C
INCHI
InChI=1S/C6H13NO2/c1-4(5(2)8)7-6(3)9/h4-5,8H,1-3H3,(H,7,9)/t4-,5-/m1/s1
FORMULA
C6H13NO2

Common name
N-[(1R,2R)-2-hydroxy-1-methyl-propyl]acetamide
IUPAC name
N-[(1R,2R)-2-hydroxy-1-methyl-propyl]acetamide
Molecular weight
131.173
clogP
-0.084
clogS
-0.528
Frequency
0.0003
HBond Acceptor
2
HBond Donor
2
Total PolarSurface Area
49.33
Number of Rings
0
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01076 | Saquinavir |
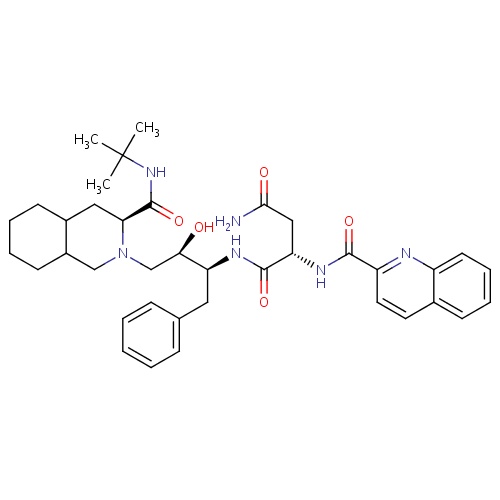
|
Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the treatment of HIV-1 with advanced immunodeficiency together with antiretroviral nucleoside analogues. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3duy_ligand_5_7071.mol2 | 3duy | 1 | -6.54 | [C@@H](NC(=O)C)([C@H](C)O)C | 9 |
| 2g94_ligand_5_6786.mol2 | 2g94 | 1 | -6.48 | C[C@H](O)[C@H](C)NC(=O)C | 9 |
| 1w6h_ligand_5_5574.mol2 | 1w6h | 1 | -6.46 | C[C@@H]([C@H](C)O)NC(=O)C | 9 |
| 3ivi_ligand_4_56.mol2 | 3ivi | 1 | -6.40 | C[C@@H](O)[C@H](C)NC(=O)C | 9 |
| 4dus_ligand_4_65.mol2 | 4dus | 1 | -6.39 | [C@H](O)([C@@H](NC(=O)C)C)C | 9 |
| 1eed_ligand_5_5382.mol2 | 1eed | 1 | -6.37 | [C@H](NC(=O)C)(C)[C@H](C)O | 9 |
| 4qgi_ligand_5_470.mol2 | 4qgi | 1 | -6.36 | CC(=O)N[C@H]([C@H](O)C)C | 9 |
203 ,
21

