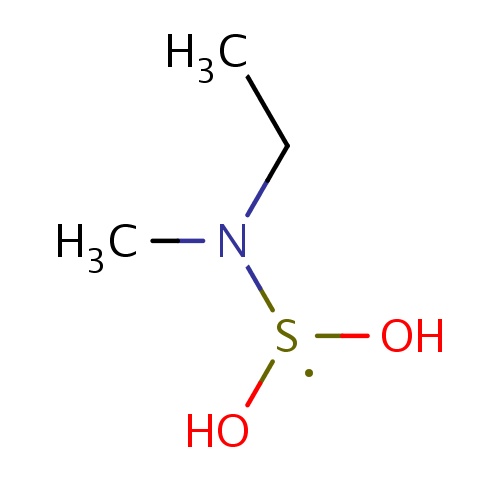
Common name
N-(dihydroxy-λ3-sulfanyl)-N-methyl-ethanamine
IUPAC name
N-(dihydroxy-λ3-sulfanyl)-N-methyl-ethanamine
SMILES
[S](O)(O)N(CC)C
Common name
N-(dihydroxy-λ3-sulfanyl)-N-methyl-ethanamine
IUPAC name
N-(dihydroxy-λ3-sulfanyl)-N-methyl-ethanamine
SMILES
[S](O)(O)N(CC)C
INCHI
InChI=1S/C3H10NO2S/c1-3-4(2)7(5)6/h5-6H,3H2,1-2H3
FORMULA
C3H10NO2S

Common name
N-(dihydroxy-λ3-sulfanyl)-N-methyl-ethanamine
IUPAC name
N-(dihydroxy-λ3-sulfanyl)-N-methyl-ethanamine
Molecular weight
124.182
clogP
-2.564
clogS
0.684
Frequency
0.0003
HBond Acceptor
3
HBond Donor
2
Total PolarSurface Area
43.7
Number of Rings
0
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
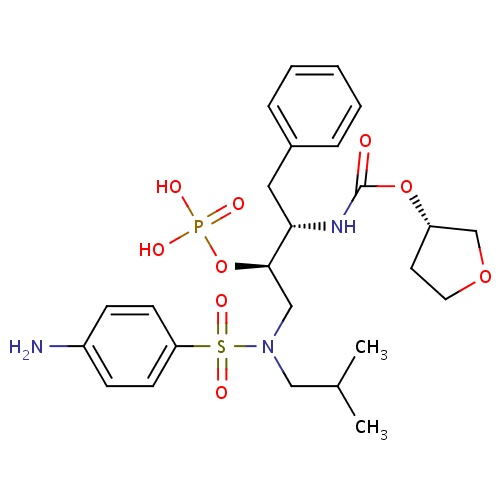
| FDBD01127 | Fosamprenavir |
|
Anti-HIV Agents; Protease Inhibitors; HIV Protease Inhibitors; Prodrugs; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; CYP3A4 Inhibitors; | Indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) infection, as well as postexposure prophylaxis of HIV infection in individuals who have had occupational or nonoccupational exposure to potentially infectious body fluids of a person known to be infected with HIV when that exposure represents a substantial risk for HIV transmission. The use of fosamprenavir is pending revision due to a potential association between the drug and myocardial infarction and dyslipidemia in HIV infected adults. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 5d1s_ligand_1_1.mol2 | 5d1s | 1 | -5.81 | N([S](O)O)(C)CC | 7 |
| 4pks_ligand_2_6.mol2 | 4pks | 1 | -5.67 | CN(CC)[S](O)O | 7 |
| 4pkt_ligand_2_4.mol2 | 4pkt | 1 | -5.63 | CN(CC)[S](O)O | 7 |
| 4pkv_ligand_2_8.mol2 | 4pkv | 1 | -5.62 | N([S](O)O)(C)CC | 7 |
| 4pkr_ligand_2_4.mol2 | 4pkr | 1 | -5.58 | N([S](O)O)(CC)C | 7 |
| 4pku_ligand_2_4.mol2 | 4pku | 1 | -5.57 | CN([S](O)O)CC | 7 |
| 3n2u_ligand_3_52.mol2 | 3n2u | 1 | -5.54 | [S](N(CC)C)(O)O | 7 |
| 5d1t_ligand_2_6.mol2 | 5d1t | 1 | -5.54 | CN(CC)[S](O)O | 7 |
145 ,
15

