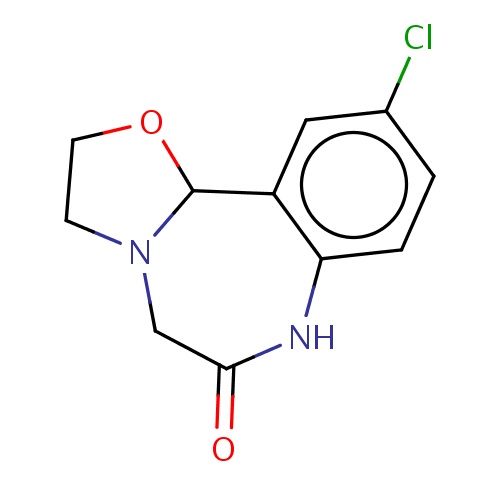
Common name
(11bR)-10-chloro-3,5,7,11b-tetrahydro-2H-oxazolo[3,2-d][1,4]benzodiazepin-6-one
IUPAC name
(11bR)-10-chloro-3,5,7,11b-tetrahydro-2H-oxazolo[3,2-d][1,4]benzodiazepin-6-one
SMILES
Clc1cc2c(cc1)NC(=O)CN3C2OCC3
Common name
(11bR)-10-chloro-3,5,7,11b-tetrahydro-2H-oxazolo[3,2-d][1,4]benzodiazepin-6-one
IUPAC name
(11bR)-10-chloro-3,5,7,11b-tetrahydro-2H-oxazolo[3,2-d][1,4]benzodiazepin-6-one
SMILES
Clc1cc2c(cc1)NC(=O)CN3C2OCC3
INCHI
InChI=1S/C11H11ClN2O2/c12-7-1-2-9-8(5-7)11-14(3-4-16-11)6-10(15)13-9/h1-2,5,11H,3-4,6H2,(H,13,15)/t11-/m1/s1
FORMULA
C11H11ClN2O2

Common name
(11bR)-10-chloro-3,5,7,11b-tetrahydro-2H-oxazolo[3,2-d][1,4]benzodiazepin-6-one
IUPAC name
(11bR)-10-chloro-3,5,7,11b-tetrahydro-2H-oxazolo[3,2-d][1,4]benzodiazepin-6-one
Molecular weight
238.670
clogP
1.547
clogS
-2.734
Frequency
0.0003
HBond Acceptor
3
HBond Donor
1
Total PolarSurface Area
41.57
Number of Rings
3
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01224 | Cloxazolam |
|
Hypnotics and Sedatives; Benzodiazepines; Neuromuscular Agents; Nervous System; Benzodiazepine Derivatives; Anxiolytics; Psycholeptics; | Used primarily as an anti-anxiety agent. Typically used short term, and may be given as a single dose of up to 100mcg/kg to reduce anxiety and tension experienced prior to surgery. |
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1t4e_ligand.mol2 | 1t4e | 0.515464 | -9.76 | Clc1ccc(cc1)[C@@H](C(=O)O)N1C(=O)c2cc(I)ccc2NC(=O)[C@@H]1c1ccc(Cl)cc1 | 33 |
| 1ezf_ligand_1_5.mol2 | 1ezf | 0.46988 | -6.84 | c12cc(ccc1N(C(=O)COC2)C)Cl | 14 |
| 1ezf_ligand_frag_0.mol2 | 1ezf | 0.46988 | -6.78 | c12cc(ccc1NC(=O)COC2)Cl | 13 |
| 1ezf_ligand_2_9.mol2 | 1ezf | 0.45614 | -7.17 | C[C@@H]1C(=O)N(c2c(cc(cc2)Cl)CO1)C | 15 |
| 1ezf_ligand_1_1.mol2 | 1ezf | 0.45614 | -7.11 | C[C@@H]1C(=O)Nc2c(cc(cc2)Cl)CO1 | 14 |
| 1inc_ligand.mol2 | 1inc | 0.452381 | -8.49 | C(=O)c1c(Cl)cccc1NC(=O)[C@H](C(C)C)NC(=O)OC(C)(C)C | 25 |
| 4r02_ligand.mol2 | 4r02 | 0.447368 | -9.51 | O=C(OCc1ccccc1)N[C@H](C(=O)N[C@H](C(=O)N[C@@H](CC(C)C)[C@H](O)C(=O)Nc1ccc(C)cc1C)CC(C)C)CC(C)C | 46 |
| 4no8_ligand.mol2 | 4no8 | 0.443243 | -9.31 | C(=O)(OCc1ccccc1)N[C@H](C(=O)N[C@H](C(=O)N[C@H]([C@H](O)C(=O)Nc1ccccc1)CC(C)C)CC(C)C)CC(C)C | 44 |
| 4gts_ligand_frag_2.mol2 | 4gts | 0.438272 | -6.12 | [S](O)(O)N1Cc2ccccc2NCC1 | 14 |

