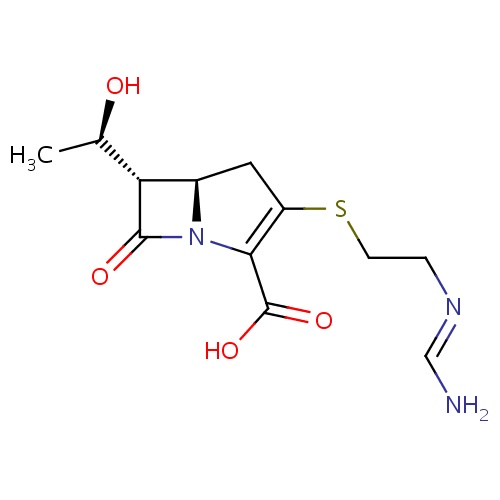
Common name
(5R)-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid
IUPAC name
(5R)-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid
SMILES
O=C1N2C(C1)CC=C2C(=O)O
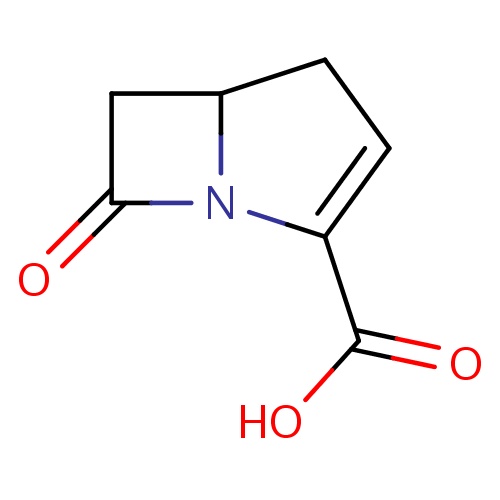
Common name
(5R)-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid
IUPAC name
(5R)-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid
SMILES
O=C1N2C(C1)CC=C2C(=O)O
INCHI
InChI=1S/C7H7NO3/c9-6-3-4-1-2-5(7(10)11)8(4)6/h2,4H,1,3H2,(H,10,11)/t4-/m1/s1
FORMULA
C7H7NO3

Common name
(5R)-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid
IUPAC name
(5R)-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid
Molecular weight
153.135
clogP
-0.190
clogS
0.357
Frequency
0.0003
HBond Acceptor
3
HBond Donor
1
Total PolarSurface Area
57.61
Number of Rings
2
Rotatable Bond
1
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2q9m_ligand.mol2 | 2q9m | 0.630435 | -6.46 | CC[C@H](C=O)[C@@H]1NC(=C2[C@@H]1CCC[C@@H]2OC)C(=O)O | 20 |
| 1nl6_ligand_1_9.mol2 | 1nl6 | 0.586777 | -5.98 | C1CC=CN(C[C@@H]1O)C(=O)C | 11 |
| 1j37_ligand_1_0.mol2 | 1j37 | 0.580645 | -6.86 | C(C)C(=O)N1[C@H](C(=O)O)CC=C1 | 12 |
| 4c1c_ligand_1_0.mol2 | 4c1c | 0.580645 | -6.23 | C(C)C(=O)N1[C@@H](C(=O)O)CC=C1 | 12 |
| 2fu8_ligand_1_0.mol2 | 2fu8 | 0.580645 | -6.13 | C(C)C(=O)N1[C@@H](C(=O)O)CC=C1 | 12 |
| 4jmx_ligand.mol2 | 4jmx | 0.577465 | -7.35 | C(=O)[C@@H]([C@@H]1NC(=C(S)C1)C(=O)O)[C@H](O)C | 16 |
| 1nl6_ligand_frag_9.mol2 | 1nl6 | 0.553719 | -5.80 | C1CC=CN(C[C@@H]1O)C=O | 10 |
| 4o4y_ligand.mol2 | 4o4y | 0.541096 | -9.38 | [NH2+]1[C@H](C(=O)N[C@H](C(=O)N2[C@H](C(=O)N[C@H](C(=O)N[C@H](C(=O)N[C@H](C(=O)NCC(=O)O)CC(C)C)CC(C)C)CCC(=O)O)CC=C2)C)CCC1 | 50 |
| 2oei_ligand.mol2 | 2oei | 0.538462 | -7.82 | [NH2+]1[C@H](C(=O)N2[C@H](C(=O)N3[C@H](C(=O)N4[C@H](C(=O)N5[C@H](C(=O)N6[C@H](C(=O)N[C@H](C(=O)N7[C@H](C(=O)N8[C@H](C(=O)O)CCC8)CCC7)CC(C)C)CCC6)CCC5)C=CC4)CCC3)CC=C2)CCC1 | 66 |
101 ,
11