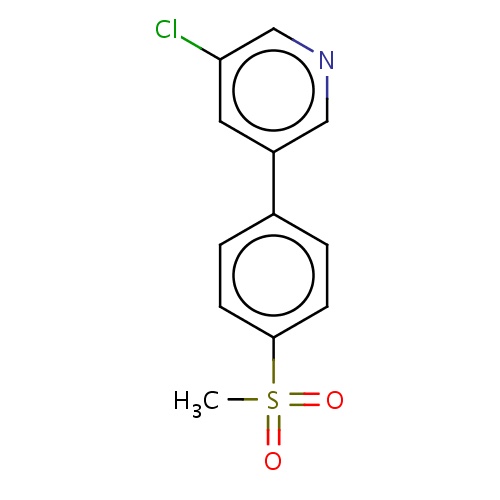
Common name
3-chloro-5-(4-methylsulfonylphenyl)pyridine
IUPAC name
3-chloro-5-(4-methylsulfonylphenyl)pyridine
SMILES
c1(ccc(cc1)S(=O)(=O)C)c2cc(cnc2)Cl
Common name
3-chloro-5-(4-methylsulfonylphenyl)pyridine
IUPAC name
3-chloro-5-(4-methylsulfonylphenyl)pyridine
SMILES
c1(ccc(cc1)S(=O)(=O)C)c2cc(cnc2)Cl
INCHI
InChI=1S/C12H10ClNO2S/c1-17(15,16)12-4-2-9(3-5-12)10-6-11(13)8-14-7-10/h2-8H,1H3
FORMULA
C12H10ClNO2S

Common name
3-chloro-5-(4-methylsulfonylphenyl)pyridine
IUPAC name
3-chloro-5-(4-methylsulfonylphenyl)pyridine
Molecular weight
267.731
clogP
2.879
clogS
-4.412
Frequency
0.0003
HBond Acceptor
3
HBond Donor
0
Total PolarSurface Area
47.03
Number of Rings
2
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01277 | Etoricoxib |

|
Musculo-Skeletal System; Antiinflammatory and Antirheumatic Products, Non-Steroids; Antiinflammatory and Antirheumatic Products; Coxibs; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of rheumatoid arthritis, osteoarthritis, ankylosing spondylitis, chronic low back pain, acute pain and gout. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 5bnj_ligand_2_1.mol2 | 5bnj | 0.563107 | -7.86 | c1(ccc(cc1)c1cc(cnc1)Cl)c1c[n+]([nH]c1)C | 19 |
| 1utr_ligand.mol2 | 1utr | 0.52809 | -8.73 | c1(c(Cl)cc(c(Cl)c1)S(=O)(=O)C)c1c(Cl)cc(c(Cl)c1)S(=O)(=O)C | 25 |
| 2cen_ligand_3_203.mol2 | 2cen | 0.52439 | -6.95 | c1cc(cnc1)c1ccc(cc1)C[NH3+] | 14 |
| 4u5l_ligand_2_0.mol2 | 4u5l | 0.52439 | -6.65 | c1cc(ccc1c1cnccc1)C[NH3+] | 14 |
| 4cpt_ligand_3_209.mol2 | 4cpt | 0.52439 | -6.60 | [NH3+]Cc1ccc(c2cccnc2)cc1 | 14 |
| 4u5o_ligand_2_0.mol2 | 4u5o | 0.52439 | -6.57 | c1(ccc(cc1)C[NH3+])c1cccnc1 | 14 |
| 1ec2_ligand_3_11.mol2 | 1ec2 | 0.518072 | -6.96 | c1(ccc(cc1)c1cccnc1)CO | 14 |
| 1ec2_ligand_3_303.mol2 | 1ec2 | 0.518072 | -6.92 | c1(ccc(cc1)c1cccnc1)CO | 14 |
| 2ohs_ligand_3_6.mol2 | 2ohs | 0.511905 | -7.18 | [NH3+]Cc1cccc(c1)c1cnccc1 | 14 |
| 2ohr_ligand_3_3.mol2 | 2ohr | 0.511905 | -7.08 | c1(cccnc1)c1cccc(c1)C[NH3+] | 14 |
102 ,
11

