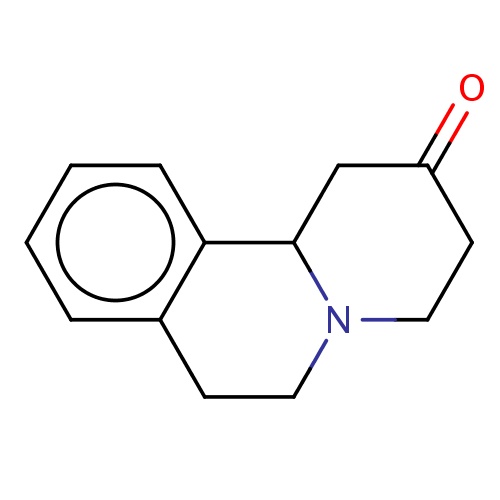
Common name
(11bS)-1,3,4,6,7,11b-hexahydropyrido[2,1-a]isoquinolin-2-one
IUPAC name
(11bS)-1,3,4,6,7,11b-hexahydropyrido[2,1-a]isoquinolin-2-one
SMILES
O=C1CCN2C(C1)c3c(cccc3)CC2
Common name
(11bS)-1,3,4,6,7,11b-hexahydropyrido[2,1-a]isoquinolin-2-one
IUPAC name
(11bS)-1,3,4,6,7,11b-hexahydropyrido[2,1-a]isoquinolin-2-one
SMILES
O=C1CCN2C(C1)c3c(cccc3)CC2
INCHI
InChI=1S/C13H15NO/c15-11-6-8-14-7-5-10-3-1-2-4-12(10)13(14)9-11/h1-4,13H,5-9H2/t13-/m0/s1
FORMULA
C13H15NO

Common name
(11bS)-1,3,4,6,7,11b-hexahydropyrido[2,1-a]isoquinolin-2-one
IUPAC name
(11bS)-1,3,4,6,7,11b-hexahydropyrido[2,1-a]isoquinolin-2-one
Molecular weight
201.264
clogP
2.488
clogS
-2.730
Frequency
0.0003
HBond Acceptor
2
HBond Donor
0
Total PolarSurface Area
20.31
Number of Rings
3
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
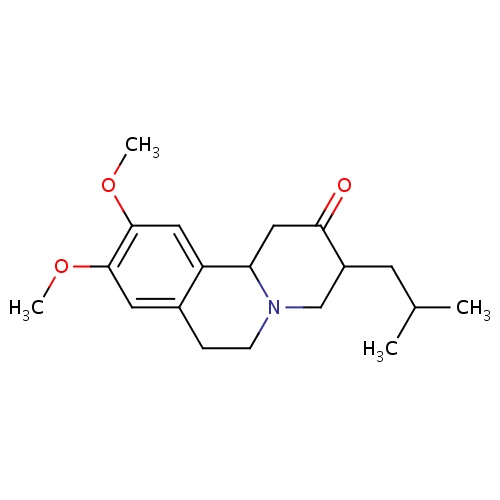
| FDBD01329 | Tetrabenazine |
|
Adrenergic Uptake Inhibitors; Nervous System; CYP2D6 Inducers; CYP2D6 Inducers (strong); | Treatment of hyperkinetic movement disorders like chorea in Huntington's disease, hemiballismus, senile chorea, Tourette syndrome and other tic disorders, and tardive dyskinesia . |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4jfi_ligand_frag_0.mol2 | 4jfi | 0.700855 | -8.12 | O=C1[C@@H]2CCC[C@H]([C@@H]3N1CCc1ccccc31)N2C(=O)C=O | 22 |
| 2buc_ligand_3_6.mol2 | 2buc | 0.69697 | -7.36 | C[C@@H]([NH3+])CC(=O)N1Cc2ccccc2CC1 | 16 |
| 4x63_ligand_1_2.mol2 | 4x63 | 0.670455 | -7.03 | C1Cc2ccccc2C[N@@H+]1C | 11 |
| 4x60_ligand_1_2.mol2 | 4x60 | 0.670455 | -6.95 | C[N@@H+]1CCc2ccccc2C1 | 11 |
| 4x61_ligand_1_2.mol2 | 4x61 | 0.670455 | -6.93 | C[N@@H+]1CCc2ccccc2C1 | 11 |
| 4np2_ligand_1_0.mol2 | 4np2 | 0.670455 | -6.33 | C1Cc2ccccc2C[N@H+]1C | 11 |
| 2f6z_ligand_2_9.mol2 | 2f6z | 0.666667 | -7.22 | C1N(Cc2ccccc2C1)C(=O)CC | 14 |
107 ,
11

