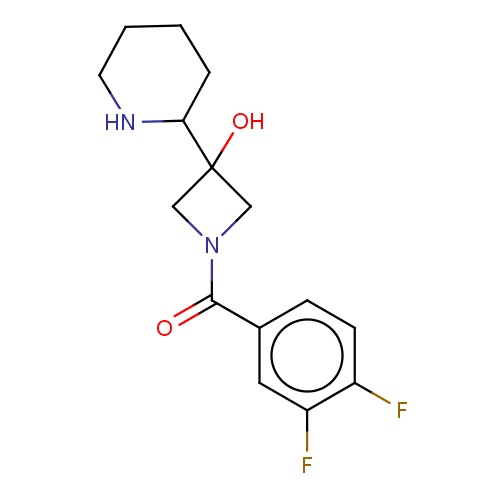
Common name
(3,4-difluorophenyl)-[3-hydroxy-3-[(2S)-2-piperidyl]azetidin-1-yl]methanone
IUPAC name
(3,4-difluorophenyl)-[3-hydroxy-3-[(2S)-2-piperidyl]azetidin-1-yl]methanone
SMILES
Fc1cc(ccc1F)C(=O)N2CC(C2)(C3NCCCC3)O
Common name
(3,4-difluorophenyl)-[3-hydroxy-3-[(2S)-2-piperidyl]azetidin-1-yl]methanone
IUPAC name
(3,4-difluorophenyl)-[3-hydroxy-3-[(2S)-2-piperidyl]azetidin-1-yl]methanone
SMILES
Fc1cc(ccc1F)C(=O)N2CC(C2)(C3NCCCC3)O
INCHI
InChI=1S/C15H18F2N2O2/c16-11-5-4-10(7-12(11)17)14(20)19-8-15(21,9-19)13-3-1-2-6-18-13/h4-5,7,13,18,21H,1-3,6,8-9H2/t13-/m0/s1
FORMULA
C15H18F2N2O2

Common name
(3,4-difluorophenyl)-[3-hydroxy-3-[(2S)-2-piperidyl]azetidin-1-yl]methanone
IUPAC name
(3,4-difluorophenyl)-[3-hydroxy-3-[(2S)-2-piperidyl]azetidin-1-yl]methanone
Molecular weight
296.312
clogP
2.558
clogS
-3.231
Frequency
0.0003
HBond Acceptor
2
HBond Donor
2
Total PolarSurface Area
52.57
Number of Rings
3
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01360 | Cobimetinib |

|
Antineoplastic Agents; | For the treatment of patients with unresectable or metastatic melanoma with a BRAF V600E or V600K mutation. Cobimetinib is used in combination with vemurafenib, a BRAF inhibitor. |
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4lmn_ligand_2_2.mol2 | 4lmn | 1 | -7.99 | c1(ccc(F)c(F)c1)C(=O)N1CC(C1)(O)[C@@H]1CCCC[NH2+]1 | 21 |
| 4lmn_ligand_1_0.mol2 | 4lmn | 0.741176 | -7.19 | c1(ccc(F)c(F)c1)C(=O)N1CC(C1)O | 15 |
| 1wbs_ligand_3_10.mol2 | 1wbs | 0.707865 | -9.07 | C(=O)(N[C@@H]1CC[C@H]2[NH2+]C[C@@H](C)[C@@H]2C1)c1cccc(F)c1 | 20 |
| 1wbs_ligand_2_5.mol2 | 1wbs | 0.707865 | -8.73 | C(=O)(N[C@@H]1CC[C@H]2[NH2+]CC[C@@H]2C1)c1cccc(F)c1 | 19 |
| 1wbv_ligand_2_2.mol2 | 1wbv | 0.707865 | -8.69 | Fc1cccc(c1)C(=O)N[C@H]1CC[C@H]2[NH2+]CC[C@@H]2C1 | 19 |
| 3cib_ligand.mol2 | 3cib | 0.669565 | -11.71 | Fc1cc(C[C@H](NC(=O)c2cc(cc(c2)C)C(=O)N(CCC)CCC)[C@H](O)[C@H]2C[C@H](CC[NH2+]2)Cc2ccccc2)cc(F)c1 | 45 |

