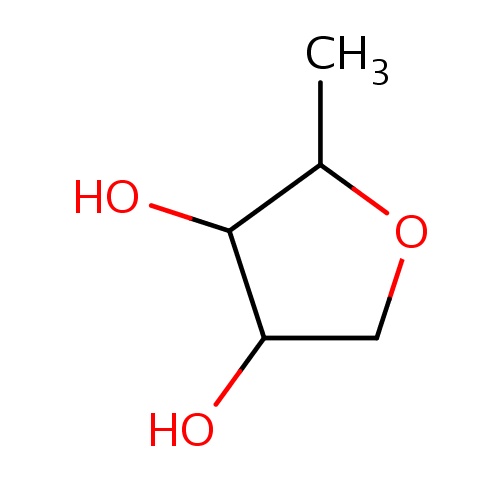
Common name
(2R,3R,4S)-2-methyltetrahydrofuran-3,4-diol
IUPAC name
(2R,3R,4S)-2-methyltetrahydrofuran-3,4-diol
SMILES
CC1OCC(C1O)O
Common name
(2R,3R,4S)-2-methyltetrahydrofuran-3,4-diol
IUPAC name
(2R,3R,4S)-2-methyltetrahydrofuran-3,4-diol
SMILES
CC1OCC(C1O)O
INCHI
InChI=1S/C5H10O3/c1-3-5(7)4(6)2-8-3/h3-7H,2H2,1H3/t3-,4+,5+/m1/s1
FORMULA
C5H10O3

Common name
(2R,3R,4S)-2-methyltetrahydrofuran-3,4-diol
IUPAC name
(2R,3R,4S)-2-methyltetrahydrofuran-3,4-diol
Molecular weight
118.131
clogP
-0.186
clogS
0.776
Frequency
0.0003
HBond Acceptor
3
HBond Donor
2
Total PolarSurface Area
49.69
Number of Rings
1
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01417 | Cangrelor |
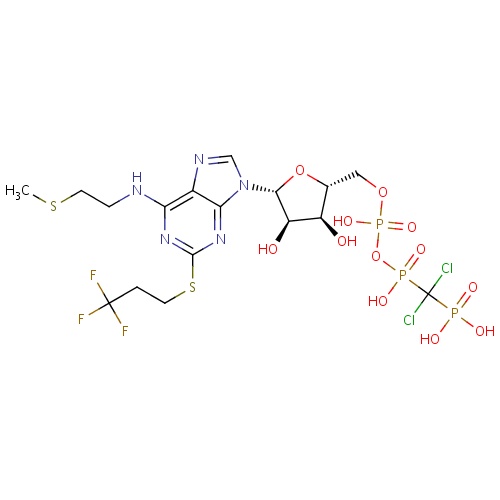
|
Platelet Aggregation Inhibitors; Antithrombotic Agents; Blood and Blood Forming Organs; Platelet Aggregation Inhibitors Excl. Heparin; Purinergic P2Y Receptor Antagonists; | For use as an adjunct to percutaneous coronary intervention (PCI) for reducing the risk of periprocedural myocardial infarction (MI), repeat coronary revascularization, and stent thrombosis (ST) in patients in who have not been treated with a P2Y12 platelet inhibitor and are not being given a glycoprotein IIb/IIIa inhibitor. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4kxl_ligand_1_3.mol2 | 4kxl | 1 | -6.67 | C[C@H]1OC[C@@H]([C@@H]1O)O | 8 |
| 2wr8_ligand_1_4.mol2 | 2wr8 | 1 | -6.65 | [C@@H]1(OC[C@@H]([C@@H]1O)O)C | 8 |
| 4kxm_ligand_1_2.mol2 | 4kxm | 1 | -6.65 | C[C@H]1OC[C@@H]([C@@H]1O)O | 8 |
| 2b1i_ligand_1_1.mol2 | 2b1i | 1 | -6.61 | C[C@H]1OC[C@@H]([C@@H]1O)O | 8 |
| 4p5d_ligand_1_2.mol2 | 4p5d | 1 | -6.59 | C[C@H]1OC[C@@H]([C@@H]1O)O | 8 |
| 2xaf_ligand_1_3.mol2 | 2xaf | 1 | -6.58 | [C@@H]1(OC[C@@H]([C@@H]1O)O)C | 8 |
| 1m9n_ligand_1_2.mol2 | 1m9n | 1 | -6.57 | [C@@H]1(OC[C@@H]([C@@H]1O)O)C | 8 |
| 4gbd_ligand_1_1.mol2 | 4gbd | 1 | -6.56 | C1[C@@H]([C@@H]([C@H](O1)C)O)O | 8 |
| 2jbv_ligand_1_3.mol2 | 2jbv | 1 | -6.55 | C[C@H]1OC[C@@H]([C@@H]1O)O | 8 |
| 1h2t_ligand_1_10.mol2 | 1h2t | 1 | -6.54 | [C@@H]1(OC[C@@H]([C@@H]1O)O)C | 8 |
636 ,
64

