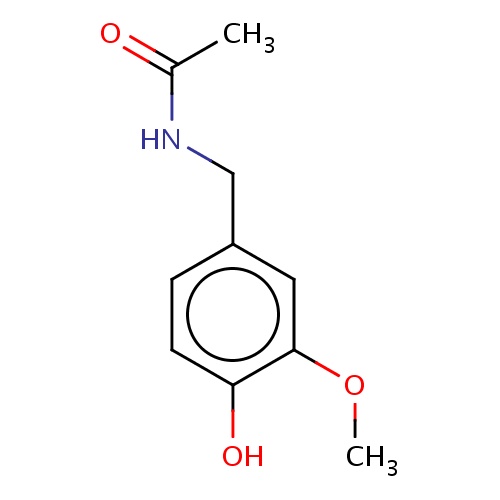
Common name
N-[(4-hydroxy-3-methoxy-phenyl)methyl]acetamide
IUPAC name
N-[(4-hydroxy-3-methoxy-phenyl)methyl]acetamide
SMILES
C(c1cc(c(cc1)O)OC)NC(=O)C
Common name
N-[(4-hydroxy-3-methoxy-phenyl)methyl]acetamide
IUPAC name
N-[(4-hydroxy-3-methoxy-phenyl)methyl]acetamide
SMILES
C(c1cc(c(cc1)O)OC)NC(=O)C
INCHI
InChI=1S/C10H13NO3/c1-7(12)11-6-8-3-4-9(13)10(5-8)14-2/h3-5,13H,6H2,1-2H3,(H,11,12)
FORMULA
C10H13NO3

Common name
N-[(4-hydroxy-3-methoxy-phenyl)methyl]acetamide
IUPAC name
N-[(4-hydroxy-3-methoxy-phenyl)methyl]acetamide
Molecular weight
195.215
clogP
1.272
clogS
-2.292
Frequency
0.0007
HBond Acceptor
3
HBond Donor
2
Total PolarSurface Area
58.56
Number of Rings
1
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01475 | Capsaicin |
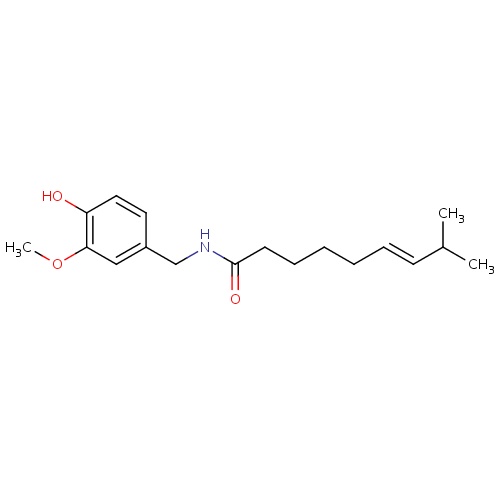
|
Anesthetics, Local; Antipruritics; Anesthetics; Sensory System Agents; Musculo-Skeletal System; Nervous System; Topical Products for Joint and Muscular Pain; Capsaicin and Similar Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; | The capsaicin 8% patch is indicated in the treatment of neuropathic pain associated with post-herpetic neuralgia. There are multiple topical capsaicin formulations available, including creams and solutions, indicated for temporary analgesia in muscle and join pain as well as neuropathic pain. |
| FDBD01692 | Zucapsaicin |
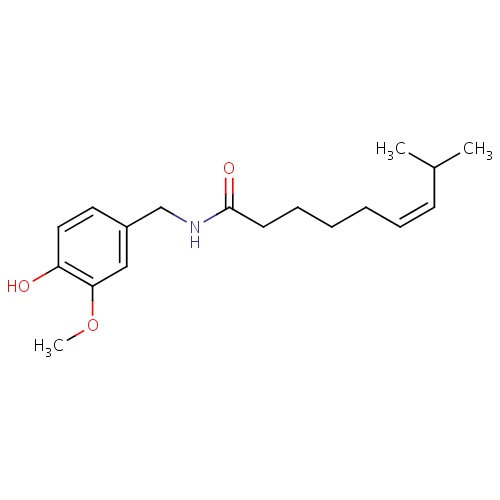
|
Musculo-Skeletal System; Topical Products for Joint and Muscular Pain; Capsaicin and Similar Agents; |
2 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4od9_ligand_4_81.mol2 | 4od9 | 0.924528 | -6.26 | C(=O)(NCc1cc(OC)ccc1)C | 13 |
| 2h96_ligand_3_15.mol2 | 2h96 | 0.867925 | -6.22 | N(C=O)Cc1cccc(c1)OC | 12 |
| 4od9_ligand_3_136.mol2 | 4od9 | 0.867925 | -5.99 | C(=O)NCc1cc(OC)ccc1 | 12 |
| 2xdl_ligand.mol2 | 2xdl | 0.85 | -7.53 | c1(c(cc(cc1)C(=O)N(CC)CC)OC)O | 17 |
| 2xdl_ligand_3_1.mol2 | 2xdl | 0.85 | -7.17 | C(C)NC(=O)c1cc(c(cc1)O)OC | 14 |
| 2xdl_ligand_3_0.mol2 | 2xdl | 0.85 | -7.15 | C(=O)(NCC)c1cc(c(cc1)O)OC | 14 |
| 1lf3_ligand_5_540.mol2 | 1lf3 | 0.85 | -6.44 | N(C(=O)c1cc(OC)c(O)cc1)CC | 14 |
| 1lf3_ligand_5_541.mol2 | 1lf3 | 0.85 | -6.34 | N(C(=O)c1ccc(O)c(OC)c1)CC | 14 |
| 4od9_ligand_4_86.mol2 | 4od9 | 0.833333 | -6.26 | C(NC(=O)C)c1ccc(OC)cc1 | 13 |
| 4gj7_ligand_4_54.mol2 | 4gj7 | 0.803279 | -7.30 | c1(cc(c(cc1)OC)O)C(=O)NC(C)C | 15 |
113 ,
12

