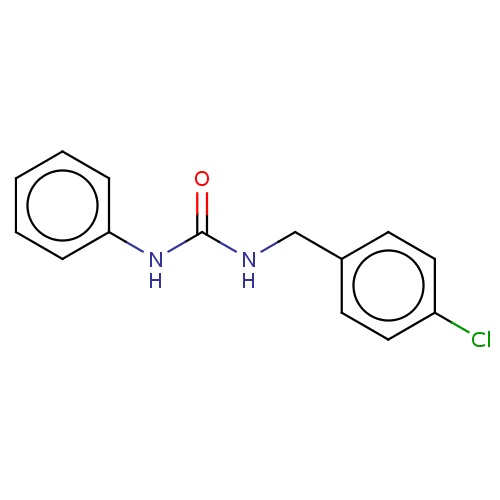
Common name
1-[(4-chlorophenyl)methyl]-3-phenylurea
IUPAC name
1-[(4-chlorophenyl)methyl]-3-phenylurea
SMILES
N(C(=O)Nc1ccccc1)Cc1ccc(cc1)Cl
Common name
1-[(4-chlorophenyl)methyl]-3-phenylurea
IUPAC name
1-[(4-chlorophenyl)methyl]-3-phenylurea
SMILES
N(C(=O)Nc1ccccc1)Cc1ccc(cc1)Cl
INCHI
InChI=1S/C14H13ClN2O/c15-12-8-6-11(7-9-12)10-16-14(18)17-13-4-2-1-3-5-13/h1-9H,10H2,(H2,16,17,18)
FORMULA
C14H13ClN2O

Common name
1-[(4-chlorophenyl)methyl]-3-phenylurea
IUPAC name
1-[(4-chlorophenyl)methyl]-3-phenylurea
Molecular weight
260.719
clogP
2.797
clogS
-4.628
Frequency
0.0003
HBond Acceptor
1
HBond Donor
2
Total PolarSurface Area
41.13
Number of Rings
2
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD03236 | pencycuron |
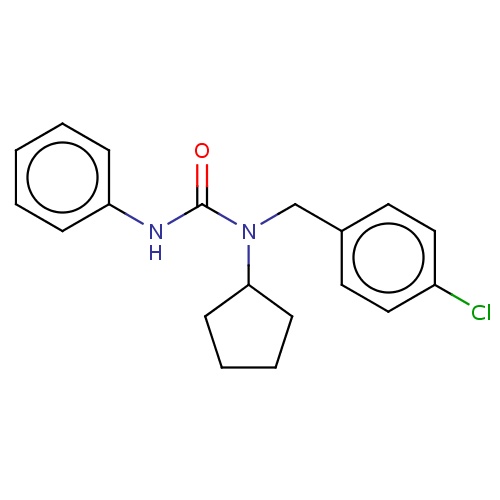
|
Fungicide | Fungicide |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4pmp_ligand_3_11.mol2 | 4pmp | 0.859375 | -8.48 | C(NC(=O)Nc1ccccc1)c1ccccc1 | 17 |
| 5bvk_ligand.mol2 | 5bvk | 0.694118 | -8.49 | c1(ccccc1NC(=O)NCc1cccnc1)Cl | 19 |
| 2q1j_ligand_3_51.mol2 | 2q1j | 0.681818 | -7.38 | CCNC(=O)Nc1ccc(cc1)Cl | 13 |
| 4mvx_ligand.mol2 | 4mvx | 0.655172 | -9.23 | O=C(NCCC[NH2+]Cc1cc(Cl)cc(Cl)c1)Nc1ccccc1 | 24 |
| 4x6n_ligand_3_23.mol2 | 4x6n | 0.647059 | -7.27 | C(=O)(NC)NCc1cccc(c1)Cl | 13 |
| 4x6m_ligand_3_46.mol2 | 4x6m | 0.647059 | -7.25 | CNC(=O)NCc1cccc(c1)Cl | 13 |
| 4hbw_ligand_frag_2.mol2 | 4hbw | 0.647059 | -6.91 | c1cc2CN(C(=O)Nc2cc1)C | 12 |
| 4hbx_ligand_frag_0.mol2 | 4hbx | 0.647059 | -6.84 | c1ccc2NC(=O)N(Cc2c1)C | 12 |
| 4hby_ligand_frag_0.mol2 | 4hby | 0.647059 | -6.78 | c1ccc2CN(C(=O)Nc2c1)C | 12 |
| 4x6n_ligand_4_39.mol2 | 4x6n | 0.637681 | -7.40 | C(=O)(NCC)NCc1cccc(c1)Cl | 14 |
105 ,
11

