Common name
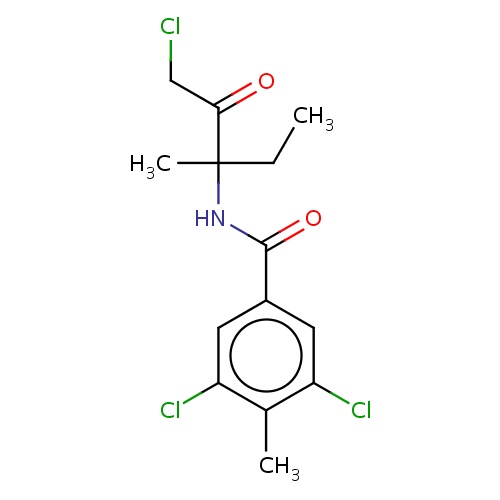
(1Z)-N-[(2R)-4-chloro-3-oxobutan-2-yl]methanimidic acid
IUPAC name
(1Z)-N-[(2R)-4-chloro-3-oxobutan-2-yl]methanimidic acid
SMILES
[C@H](C)(NC=O)C(=O)CCl
Common name
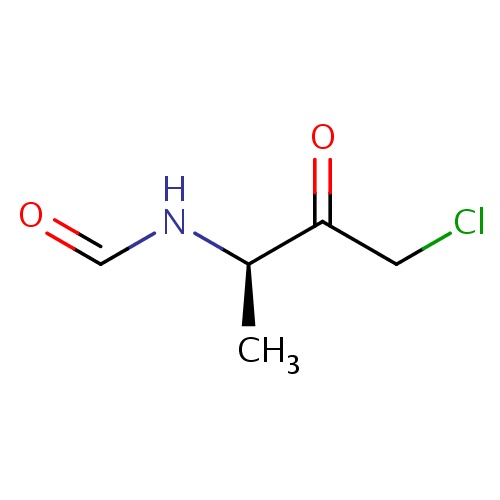
(1Z)-N-[(2R)-4-chloro-3-oxobutan-2-yl]methanimidic acid
IUPAC name
(1Z)-N-[(2R)-4-chloro-3-oxobutan-2-yl]methanimidic acid
SMILES
[C@H](C)(NC=O)C(=O)CCl
INCHI
InChI=1S/C5H8ClNO2/c1-4(7-3-8)5(9)2-6/h3-4H,2H2,1H3,(H,7,8)/t4-/m1/s1
FORMULA
C5H8ClNO2

Common name
(1Z)-N-[(2R)-4-chloro-3-oxobutan-2-yl]methanimidic acid
IUPAC name
(1Z)-N-[(2R)-4-chloro-3-oxobutan-2-yl]methanimidic acid
Molecular weight
149.576
clogP
1.119
clogS
-0.784
Frequency
0.0003
HBond Acceptor
3
HBond Donor
1
Total PolarSurface Area
49.66
Number of Rings
0
Rotatable Bond
3
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4ps0_ligand_2_102.mol2 | 4ps0 | 0.633333 | -5.42 | C(/N=C\O)C(=O)C | 7 |
| 3avz_ligand_2_102.mol2 | 3avz | 0.566667 | -5.48 | [C@H](C=O)(/N=C\O)C | 7 |
| 4q2k_ligand_2_3.mol2 | 4q2k | 0.566667 | -5.39 | C(=N\[C@H](C=O)C)\O | 7 |
| 3hd3_ligand_4_260.mol2 | 3hd3 | 0.5 | -5.64 | C[C@H](CC)/N=C\O | 7 |
| 3hd3_ligand_4_204.mol2 | 3hd3 | 0.5 | -5.59 | CC[C@H](C)/N=C\O | 7 |
| 3avz_ligand_1_12.mol2 | 3avz | 0.5 | -5.03 | C(C=O)/N=C\O | 6 |
| 4q2k_ligand_1_1.mol2 | 4q2k | 0.5 | -4.98 | C(=N\CC=O)\O | 6 |
| 4gm8_ligand_3_196.mol2 | 4gm8 | 0.483871 | -5.86 | C(C)C(CC)/N=C\O | 8 |
| 3hd3_ligand_5_336.mol2 | 3hd3 | 0.483871 | -5.73 | CCC(CC)/N=C\O | 8 |
| 1w6h_ligand_4_1695.mol2 | 1w6h | 0.470588 | -6.22 | C[C@@H]([C@H](C)O)/N=C\O | 8 |
107 ,
11