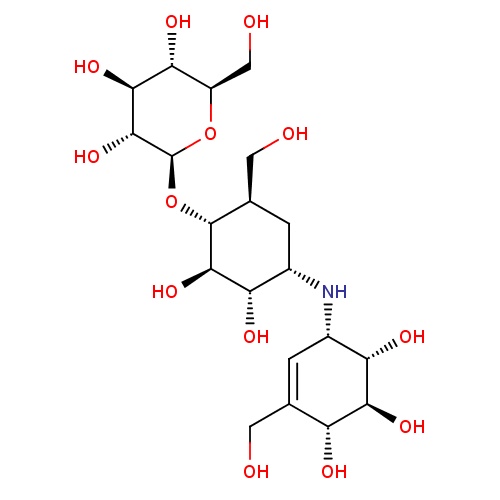
Common name
validamycin
Formula
C20H35NO13
CAS
37248-47-8
IUPAC name
(1R,2R,3S,4S,6R)-2,3-dihydroxy-6-hydroxymethyl-4-[(1S,4R,5S,6S)-4,5,6-trihydroxy-3-hydroxymethylcyclohex-2-enylamino]cyclohexyl ?-D-glucopyranoside
SMILES
C1=C(CO)[C@H]([C@@H]([C@H]([C@H]1N[C@H]1C[C@H](CO)[C@H]([C@@H]([C@H]1O)O)O[C@H]1[C@@H]([C@H]([C@@H]([C@@H](CO)O1)O)O)O)O)O)O
INCHI
InChI=1S/C20H35NO13/c22-3-6-1-8(12(26)15(29)11(6)25)21-9-2-7(4-23)19(17(31)13(9)27)34-20-18(32)16(30)14(28)10(5-24)33-20/h1,7-32H,2-5H2/t7-,8+,9+,10-,11-,12+,13+,14-,15+,16+,17-,18-,19-,20+/m1/s1
Physicochemical Properties
Molecular weight:
Molar refractivity:
Num. bonds:
Num. double bonds:
Num. triple bonds:
Num. heavy atoms:
Num. nitrogen atom:
Num. oxygen atoms:
Num. sulphur atoms :
Num. P atoms:
Num. halogen atoms:
Num. H-bond acc:
Num. H-bond donors:
Complexity of system:
Lipophilicity
ALogP:
MLogP:
XLogP:
Water Solubility
CLogS:
Geometric
PPSA-1:
PNSA-1:
RPCS:
RNCS:
THSA:
TPSA:
RHSA:
RPSA:
Electrostatic
RPCG:
RNCG:
Flexibility
Num. Rot. bonds: 7
Frac. of Rot. bonds:
AgroPAD
Photostability
Num. arom. bonds:
Num. arom. atoms:
Num. arom. rings:
Num. rings:
Topological
topoRadius:
topoDiameter:
TopoPSA:
Fungicide-likeness scores
The higher the score, the better the fungicide-likeness
RDL:
QEF:
Gau:
