Help
1.Introduction
InsectiPAD(Insecticide Physicochemical-properties Analysis Database) is a special agrochemical database to not only provide information about insecticides but also qualitative and quantitative evaluate insecticide-likeness of small molecules. It displays the information on all approved insecticides including their CAS numbers, common names, 3D structures and physicochemical properties. This website also allows you to compute physicochemical descriptors and insecticide-likeness scores of any small molecule to support agrochemical discovery.
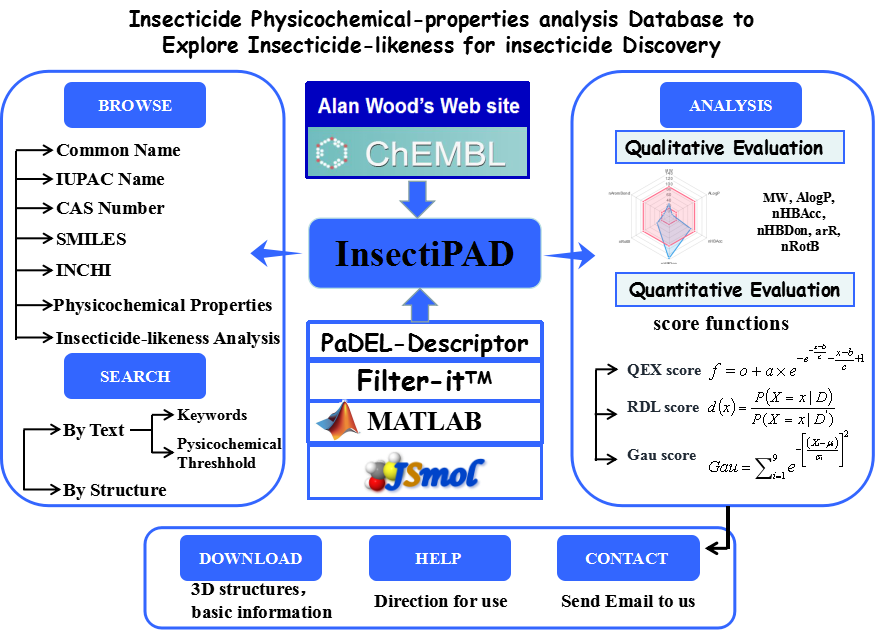
Fig 1. The Architecture of InsectiPAD
2.Browser Recommendation
We tested our database using different browsers on different systems (IE10 or later on Windows, Firefox on Windows, Mac OS and Linux, Google Chrome on Windows, Mac OS, and Linux, Apple Safari on Windows and Mac os) to assure the normal display. The testing results showed good compatibility.
3.Browse Method
You can browse structures and physicochemical properties of these molecules directly in the Browse page, click ID-link to view more basic information(common name, IUPAC name, CAS number, smiles, inchi), physicochemical properties(MW, ALogP, AMR, nAromBond, CLogS, TopoPSA, nHBAcc, nHBDon etc.), qualitative analysis of the distribution of molecular physicochemical properties and quantitative analysis of insecticide-likeness scores.
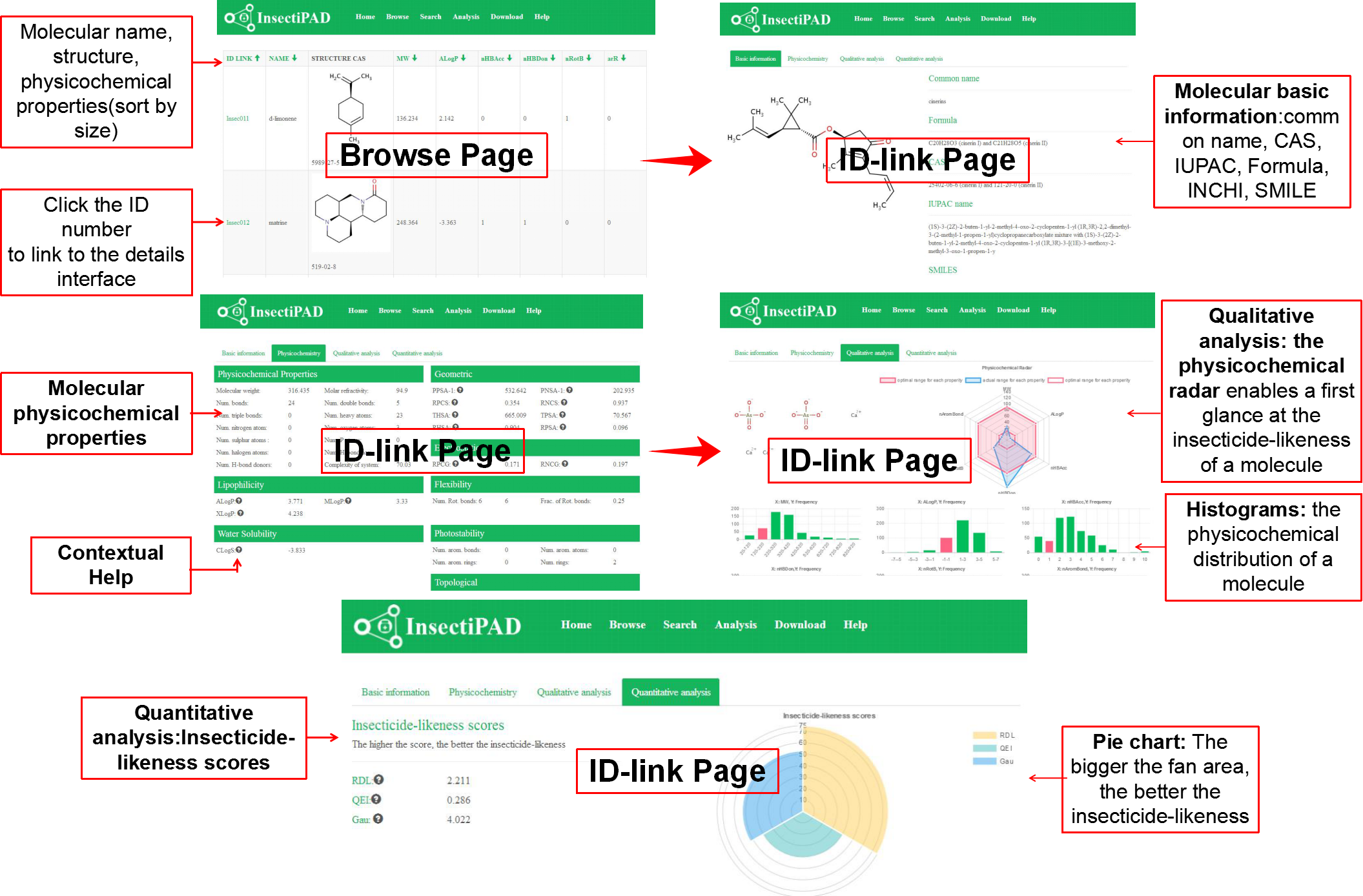
Fig 2. Browse InsectiPAD
4.Search Method
Insecticides can be retrieved through both a text-based keyword search and a structure-based similarity search. For text-based search, insecticides match the keywords will be showed. You can also refine your search by choosing some specific physicochemical characteristics for the compounds. For structure-based search, insecticide with similar structure with your input structure will be presented.
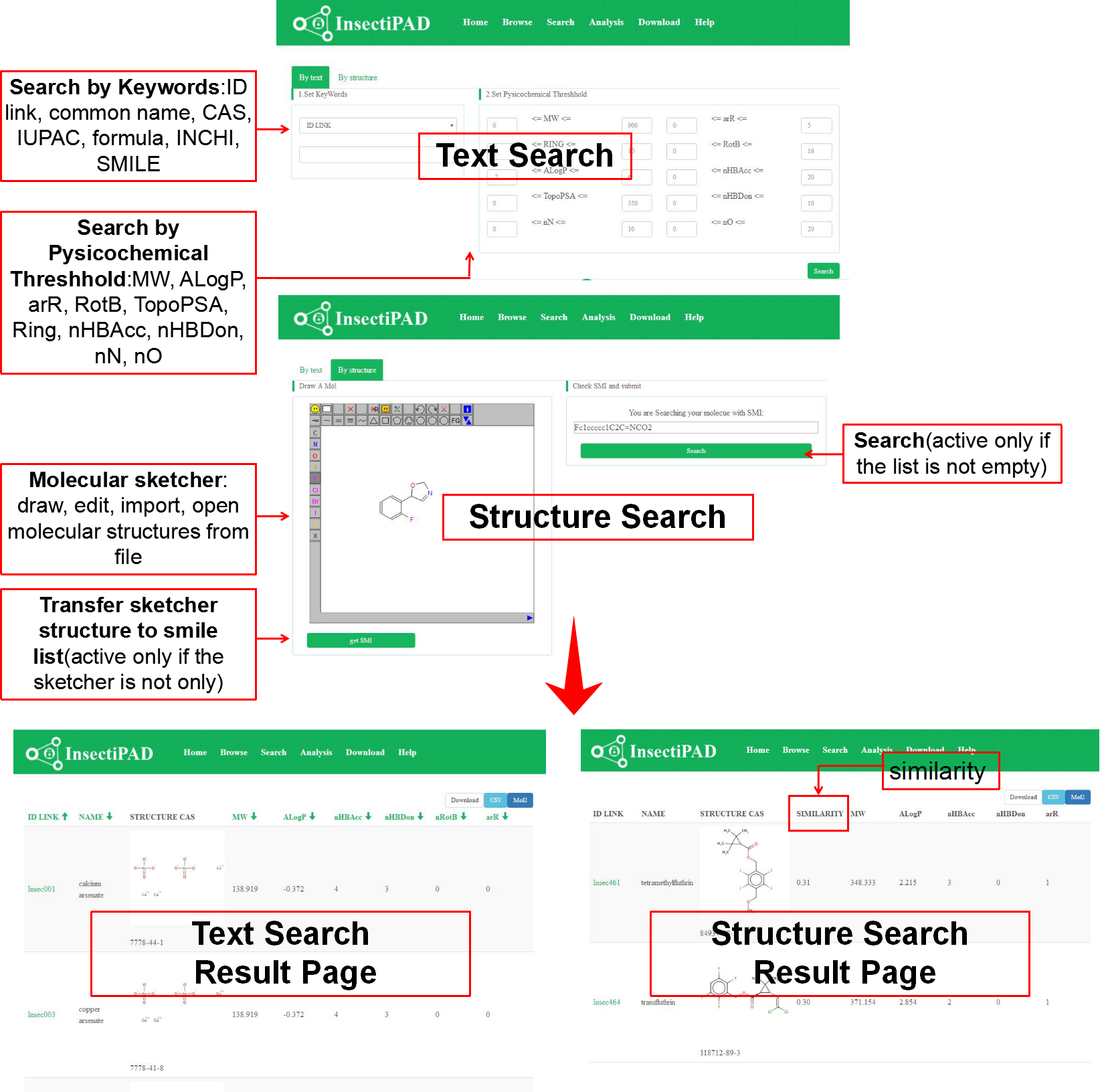
Fig 3. Search InsectiPAD
5.Analysis Method
Predicting the insecticide-likeness of experimental molecules is important in the discovery process. Our database can help you to analysis the insecticide-likeness of your compounds. The input is an user-defined structure generated by JSME. Your selected compounds with related physicochemical properties and their distribution histograms, bioavailability radars and insecticide-likeness scores will be showed in a few minute. You can evaluate the insecticide-likeness of compounds by the Radar (the pink area represents the optimal range for each properties) and histograms at a first glance or according to the insecticide-likeness scores (The higher the score, the better the pesticide-likeness of your compounds). These evaluating methods are not meant to be as accurate as virtual screening tools, but the results are indicators of compounds showing desirable insecticide-likeness physicochemical properties.
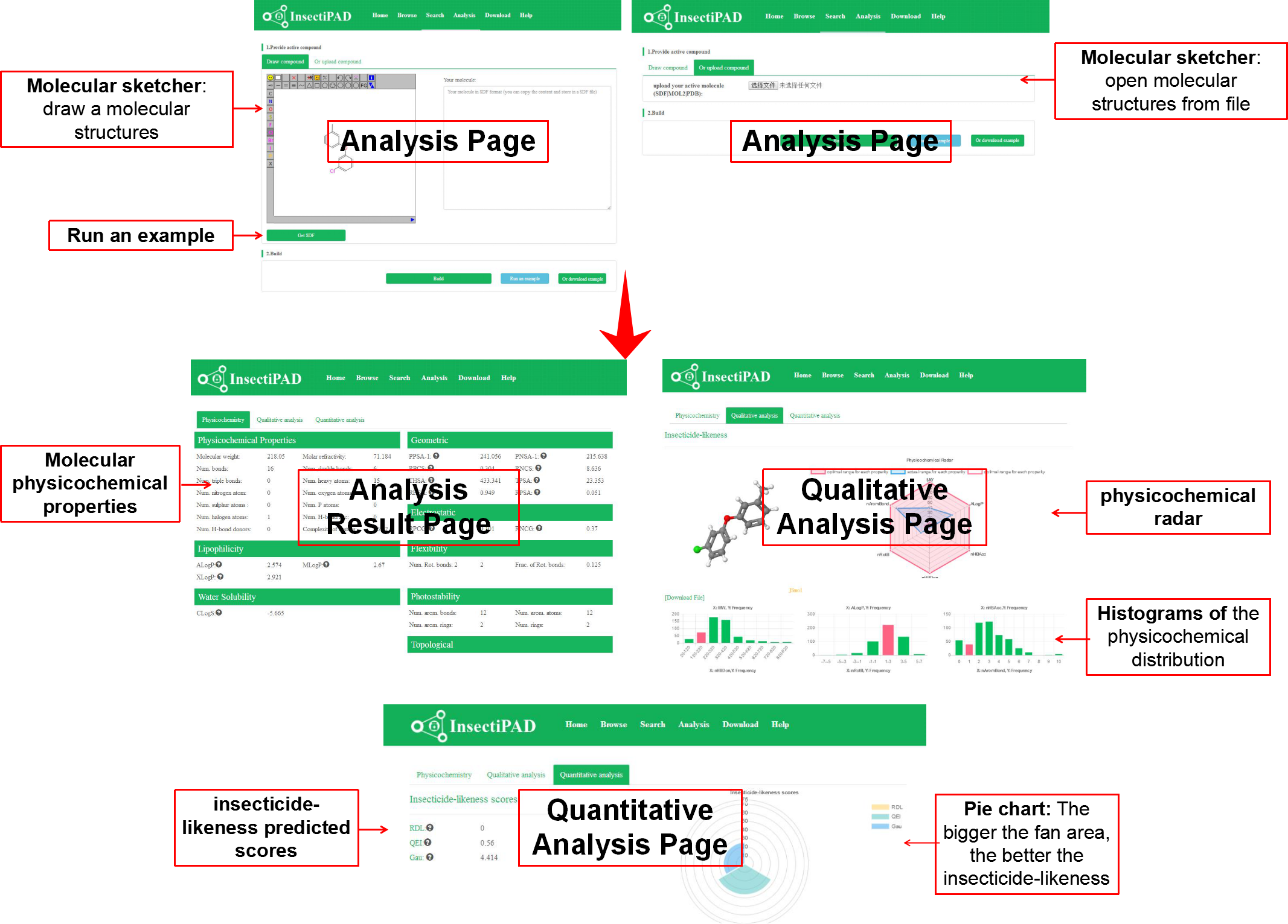
Fig 4. Analysis InsectiPAD
6.Score Function Building
We establish quantitative assessments (QEI, RDL and Gau) to efficient prioritize insecticide-likeness compounds. Every approach relies on a small number of molecular descriptors which are relevant, accessible and easy to compute to describe the distribution of a set of molecules.
1.QEI accurately describe six molecular properties(MW, ALogP, HBA, HBD, nRotB, arR) over the all approved insecticides, parameterized by a,b,c,o coefficients computed for each distribution of insecticides properties. And the individual dfi (i molecular descriptor) was joined by computing geometric means and logarithm.

2.Following bayesian probability theory, we randomly selected 1000 compounds from the ChEMBL database considered as a negative set, and pesticides as positive sets to build RDL function. P(X|D) is the probability of the property X given that a compound is a drug. P(X|D') the probability of the property X given that a compound not being a drug. We considered six properties(MW, LogP, HBA, HBD, nRotB, arR) used in QEI.

3.Gaussian function describes a more multivariate descriptors' distribution to ascertain insecticide likeness of small molecule compounds, including MW, LogP, HBA, HBD, PSA, nRing, nN, nO and nRotB. The values Xi represent the nine physicochemical properties respectively. The parameters μi and σi represent the average and standard deviation of each Xi.