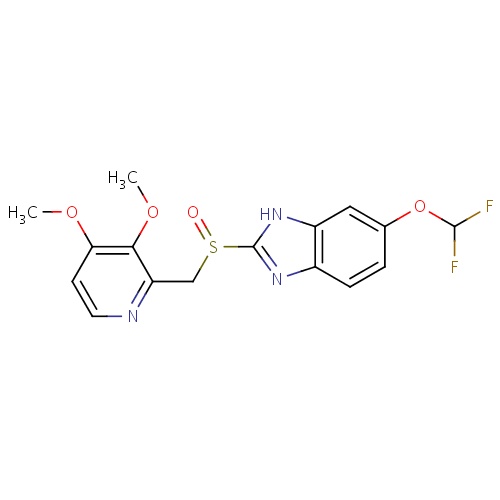
IUPAC name
6-(difluoromethoxy)-2-[(3,4-dimethoxypyridin-2-yl)methanesulfinyl]-1H-1,3-benzodiazole
SMILES
COC1=C(OC)C(CS(=O)C2=NC3=C(N2)C=C(OC(F)F)C=C3)=NC=C1
Compound class
Anti-Ulcer Agents; Proton Pump Inhibitors; Alimentary Tract and Metabolism; Drugs for Peptic Ulcer and Gastro-Oesophageal Reflux Disease (Gord); Drugs for Acid Related Disorders; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors;
Therapeutic area
Short-term (up to 16 weeks) treatment of erosive esophagitis.
Common name
Pantoprazole
IUPAC name
6-(difluoromethoxy)-2-[(3,4-dimethoxypyridin-2-yl)methanesulfinyl]-1H-1,3-benzodiazole
SMILES
COC1=C(OC)C(CS(=O)C2=NC3=C(N2)C=C(OC(F)F)C=C3)=NC=C1
INCHI
InChI=1S/C16H15F2N3O4S/c1-23-13-5-6-19-12(14(13)24-2)8-26(22)16-20-10-4-3-9(25-15(17)18)7-11(10)21-16/h3-7,15H,8H2,1-2H3,(H,20,21)
FORMULA
C16H15F2N3O4S

Common name
Pantoprazole
IUPAC name
6-(difluoromethoxy)-2-[(3,4-dimethoxypyridin-2-yl)methanesulfinyl]-1H-1,3-benzodiazole
Molecular weight
383.370
clogP
2.623
clogS
-5.261
HBond Acceptor
6
HBond Donor
1
Total Polar Surface Area
86.33
Number of Rings
3
Rotatable Bond
7
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
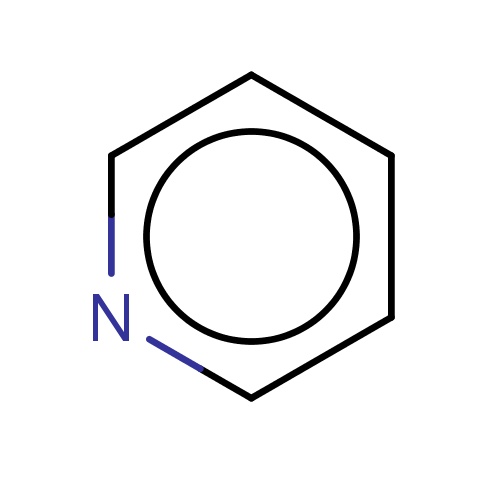
| FDBF00014 | pyridine |
|
c1cccnc1 | 0.0333 |
| FDBF00356 | hydroxysulfanylmethane |

|
S(O)C | 0.0069 |
| FDBF00359 | difluoromethanol |
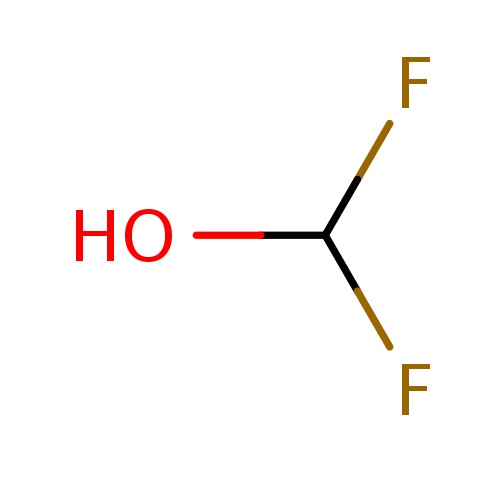
|
FC(F)O | 0.0058 |
| FDBF00364 | 3,4-dimethoxypyridine |
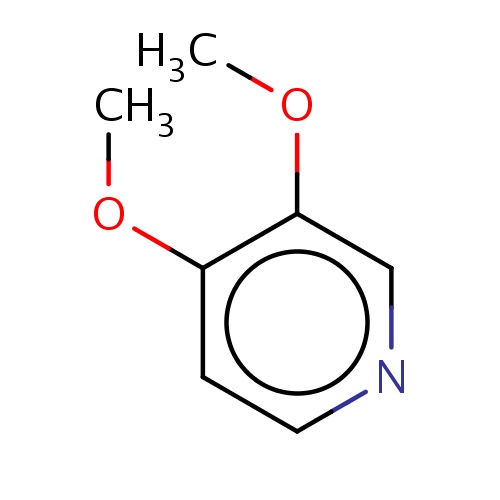
|
O(C)c1cnccc1OC | 0.0003 |
| FDBF00368 | 3,4-dimethoxy-2-methyl-pyridine |
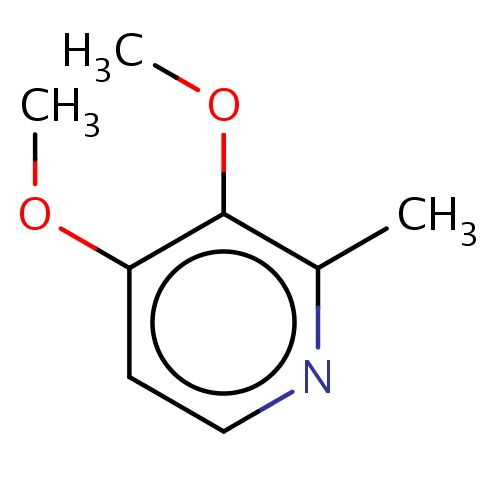
|
O(C)c1c(nccc1OC)C | 0.0003 |
| FDBF00370 | difluoromethane |

|
FCF | 0.0151 |
| FDBF00371 | benzimidazole |
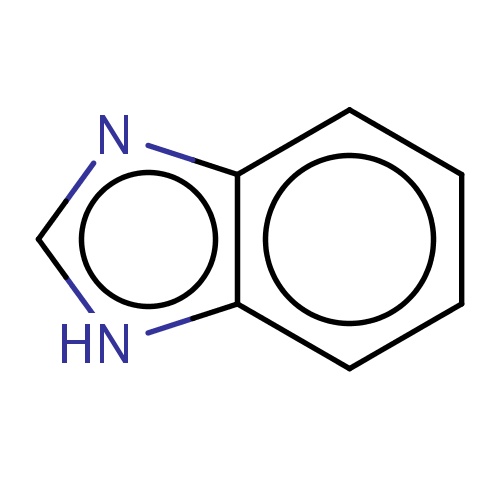
|
[nH]1c2c(nc1)cccc2 | 0.0045 |

