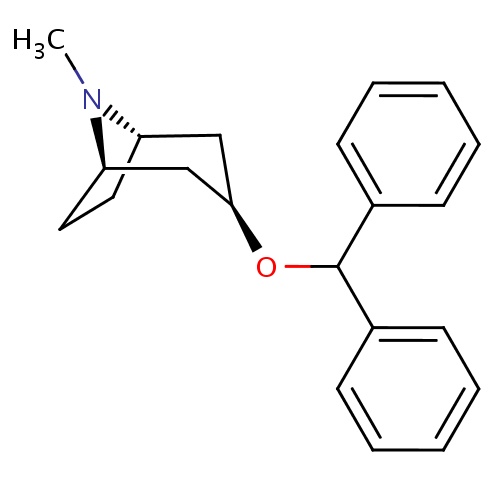
IUPAC name
(1R,3R,5S)-3-(diphenylmethoxy)-8-methyl-8-azabicyclo[3.2.1]octane
SMILES
[H][C@]12CC[C@]([H])(C[C@@]([H])(C1)OC(C1=CC=CC=C1)C1=CC=CC=C1)N2C
Compound class
Dopamine Uptake Inhibitors; Antiparkinson Agents; Muscarinic Antagonists; Parasympatholytics; Antidyskinetics; Nervous System; Ethers of Tropine or Tropine Derivatives; Anti-Parkinson Drugs; Anticholinergics; Cytochrome P-450 CYP2C19 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong);
Therapeutic area
For use as an adjunct in the therapy of all forms of parkinsonism and also for use in the control of extrapyramidal disorders due to neuroleptic drugs.
Common name
Benzatropine
IUPAC name
(1R,3R,5S)-3-(diphenylmethoxy)-8-methyl-8-azabicyclo[3.2.1]octane
SMILES
[H][C@]12CC[C@]([H])(C[C@@]([H])(C1)OC(C1=CC=CC=C1)C1=CC=CC=C1)N2C
INCHI
InChI=1S/C21H25NO/c1-22-18-12-13-19(22)15-20(14-18)23-21(16-8-4-2-5-9-16)17-10-6-3-7-11-17/h2-11,18-21H,12-15H2,1H3/t18-,19+,20+
FORMULA
C21H25NO

Common name
Benzatropine
IUPAC name
(1R,3R,5S)-3-(diphenylmethoxy)-8-methyl-8-azabicyclo[3.2.1]octane
Molecular weight
307.429
clogP
3.844
clogS
-4.146
HBond Acceptor
2
HBond Donor
0
Total Polar Surface Area
12.47
Number of Rings
4
Rotatable Bond
4
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
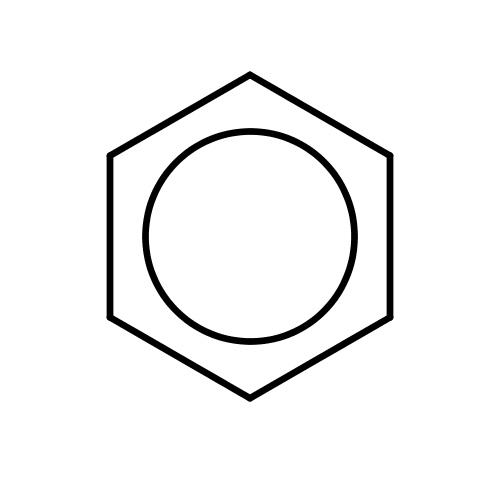
| FDBF00005 | benzene |
|
c1ccccc1 | 0.2824 |
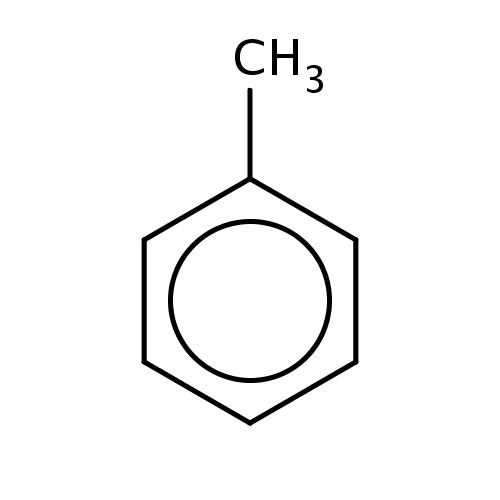
| FDBF00023 | toluene |
|
c1(ccccc1)C | 0.1268 |
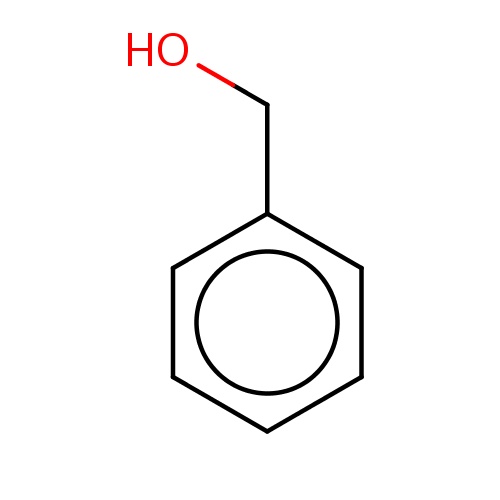
| FDBF00304 | phenylmethanol |
|
c1(ccccc1)CO | 0.0168 |
| FDBF00503 | (1R,5S)-8-methyl-8-azabicyclo[3.2.1]octan-3-ol |
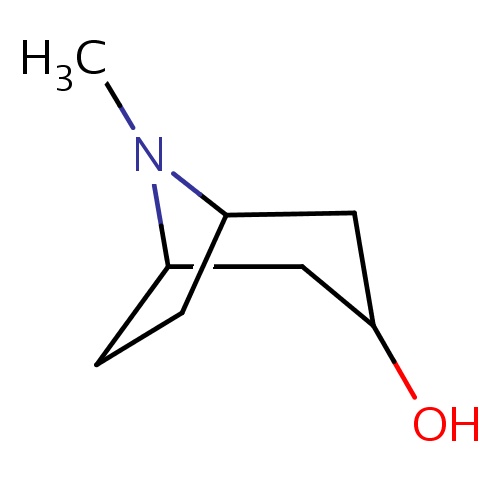
|
OC1CC2N(C(C1)CC2)C | 0.0003 |
| FDBF00504 | (1R,5S)-3-methoxy-8-methyl-8-azabicyclo[3.2.1]octane |
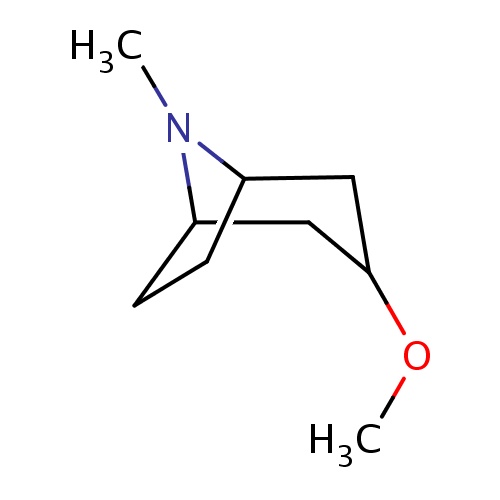
|
O(C)C1CC2N(C(C1)CC2)C | 0.0003 |
| FDBF00505 | (1R,5S)-3-benzyloxy-8-methyl-8-azabicyclo[3.2.1]octane |
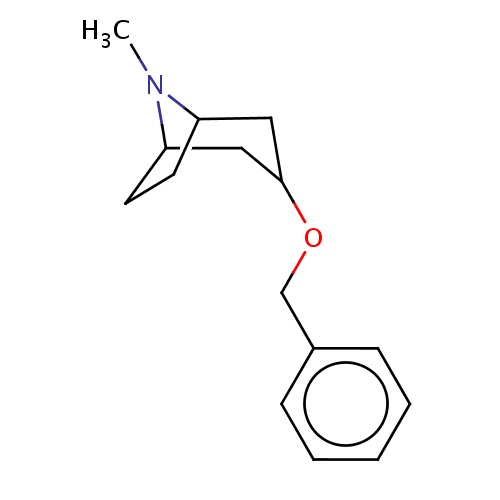
|
c1(ccccc1)COC2CC3N(C(C2)CC3)C | 0.0003 |
| FDBF00506 | (1S,5R)-8-methyl-8-azabicyclo[3.2.1]octane |

|
C12CCCC(N1C)CC2 | 0.0024 |

