IUPAC name
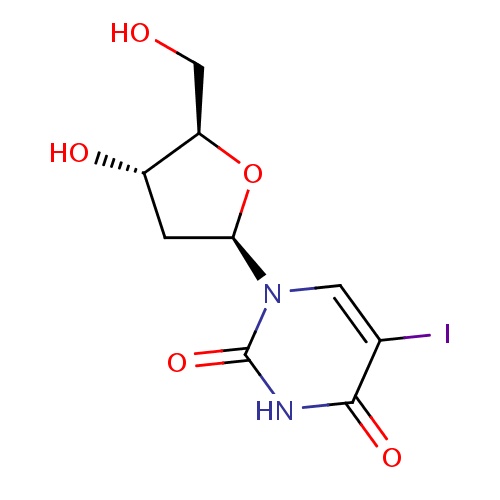
1-[(2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-iodo-1,2,3,4-tetrahydropyrimidine-2,4-dione
SMILES
OC[C@H]1O[C@H](C[C@@H]1O)N1C=C(I)C(=O)NC1=O
Compound class
Antiviral Agents; Nucleic Acid Synthesis Inhibitors; Ophthalmologicals; Sensory Organs; Antiinfectives for Systemic Use; Dermatologicals; Direct Acting Antivirals; Antivirals for Systemic Use; Nucleosides and Nucleotides Excl. Reverse Transcriptase Inhibitors; Antiinfectives;
Therapeutic area
For use in keratoconjunctivitis and keratitis caused by herpes simplex virus.
Common name
Idoxuridine
IUPAC name
1-[(2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-iodo-1,2,3,4-tetrahydropyrimidine-2,4-dione
SMILES
OC[C@H]1O[C@H](C[C@@H]1O)N1C=C(I)C(=O)NC1=O
INCHI
InChI=1S/C9H11IN2O5/c10-4-2-12(9(16)11-8(4)15)7-1-5(14)6(3-13)17-7/h2,5-7,13-14H,1,3H2,(H,11,15,16)/t5-,6+,7+/m0/s1
FORMULA
C9H11IN2O5

Common name
Idoxuridine
IUPAC name
1-[(2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-iodo-1,2,3,4-tetrahydropyrimidine-2,4-dione
Molecular weight
354.099
clogP
0.472
clogS
-0.811
HBond Acceptor
5
HBond Donor
3
Total Polar Surface Area
104.55
Number of Rings
2
Rotatable Bond
2