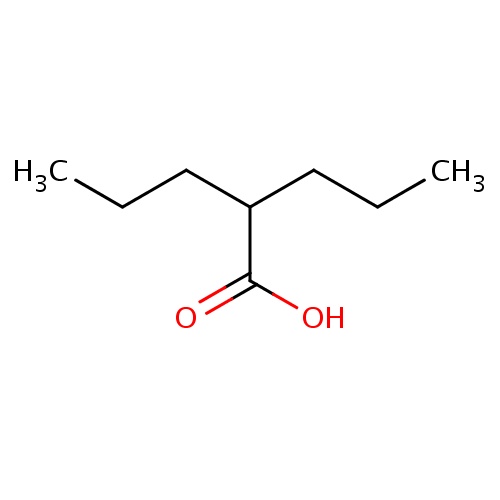
IUPAC name
2-propylpentanoic acid
SMILES
CCCC(CCC)C(O)=O
Compound class
Enzyme Inhibitors; Anticonvulsants; Antimanic Agents; GABA Agents; Nervous System; Antiepileptics; Fatty Acid Derivatives; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2B6 Inhibitors (strong); CYP3A4 Inhibitors;
Therapeutic area
For treatment and management of seizure disorders, mania, and prophylactic treatment of migraine headache. In epileptics, valproic acid is used to control absence seizures, tonic-clonic seizures (grand mal), complex partial seizures, and the seizures associated with Lennox-Gastaut syndrome.
Common name
Valproic Acid
IUPAC name
2-propylpentanoic acid
SMILES
CCCC(CCC)C(O)=O
INCHI
InChI=1S/C8H16O2/c1-3-5-7(6-4-2)8(9)10/h7H,3-6H2,1-2H3,(H,9,10)
FORMULA
C8H16O2

Common name
Valproic Acid
IUPAC name
2-propylpentanoic acid
Molecular weight
144.211
clogP
1.600
clogS
-1.672
HBond Acceptor
2
HBond Donor
1
Total Polar Surface Area
37.3
Number of Rings
0
Rotatable Bond
5
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
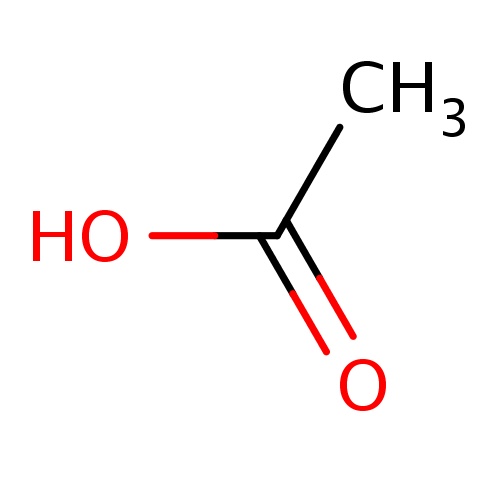
| FDBF00004 | acetic acid |
|
CC(=O)O | 0.0687 |
| FDBF00007 | propane |

|
C(C)C | 0.2412 |
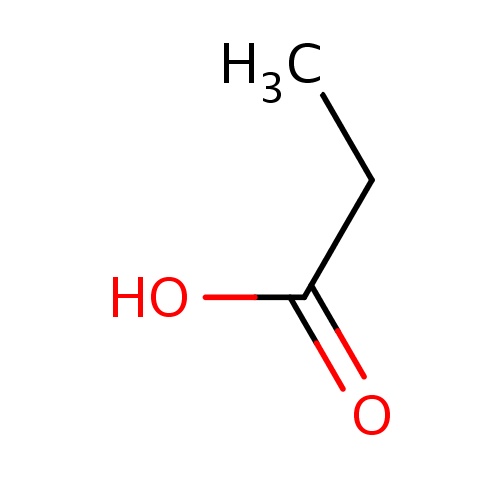
| FDBF00047 | propionic acid |
|
CCC(=O)O | 0.0395 |
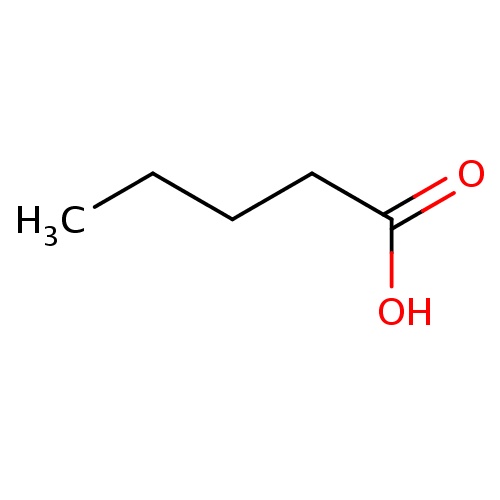
| FDBF00062 | pentanoic acid |
|
CCCCC(=O)O | 0.0079 |
| FDBF00240 | 2-methylpropanoic acid |
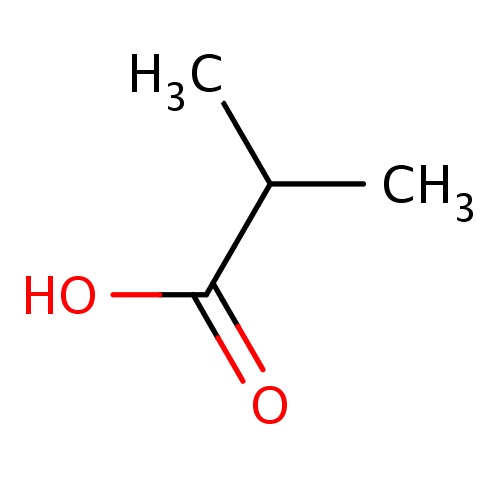
|
CC(C)C(=O)O | 0.0027 |
| FDBF00699 | (2S)-2-methylpentanoic acid |

|
C(C)CC(C)C(=O)O | 0.0007 |
| FDBF00700 | (2R)-2-methylpentanoic acid |

|
C(C)CC(C)C(=O)O | 0.0007 |

