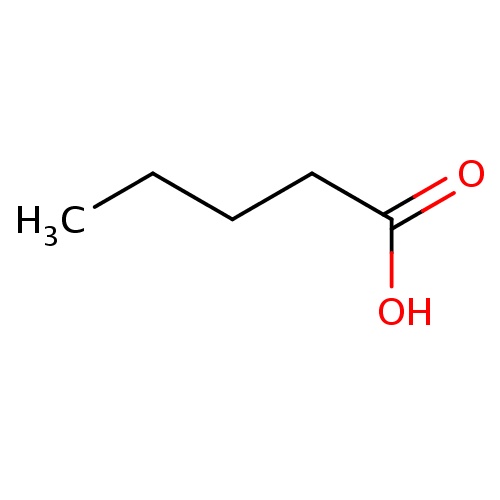
Common name
pentanoic acid
IUPAC name
pentanoic acid
SMILES
CCCCC(=O)O
Common name
pentanoic acid
IUPAC name
pentanoic acid
SMILES
CCCCC(=O)O
INCHI
InChI=1S/C5H10O2/c1-2-3-4-5(6)7/h2-4H2,1H3,(H,6,7)
FORMULA
C5H10O2

Common name
pentanoic acid
IUPAC name
pentanoic acid
Molecular weight
102.132
clogP
0.522
clogS
-0.780
Frequency
0.0079
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
37.3
Number of Rings
0
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00014 | Biotin |
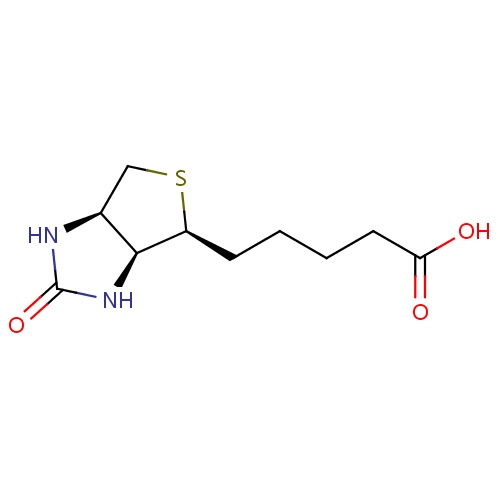
|
Vitamin B Complex; Vitamins; Dietary Supplements; Micronutrients; Supplements; Alimentary Tract and Metabolism; | For nutritional supplementation, also for treating dietary shortage or imbalance. |
| FDBD00024 | Alpha-Linolenic Acid |
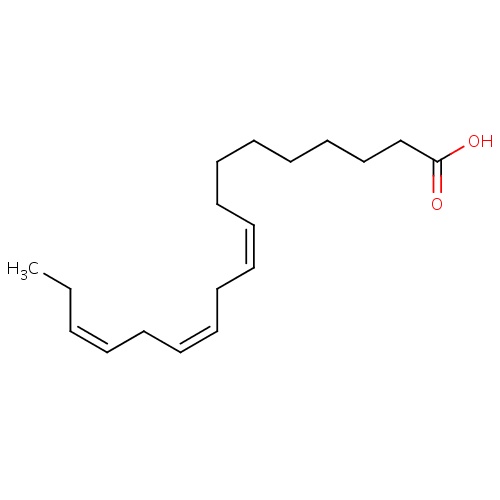
|
Dietary Supplements; Micronutrients; Supplements; | For nutritional supplementation and for treating dietary shortage or imbalance. |
| FDBD00045 | Dihomo-gamma-linolenic acid |
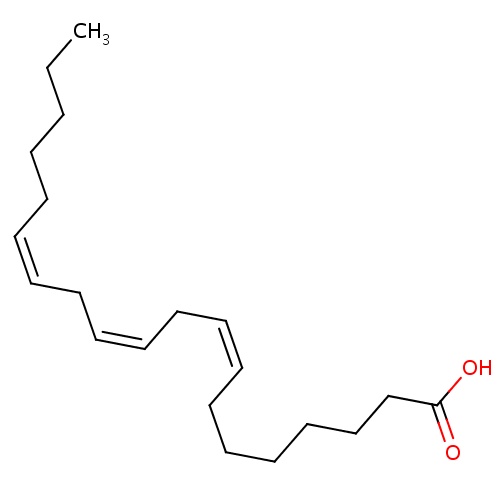
|
Dietary Supplements; Micronutrients; Supplements; | For nutritional supplementation, also for treating dietary shortage or imbalance. |
| FDBD00055 | Lipoic Acid |
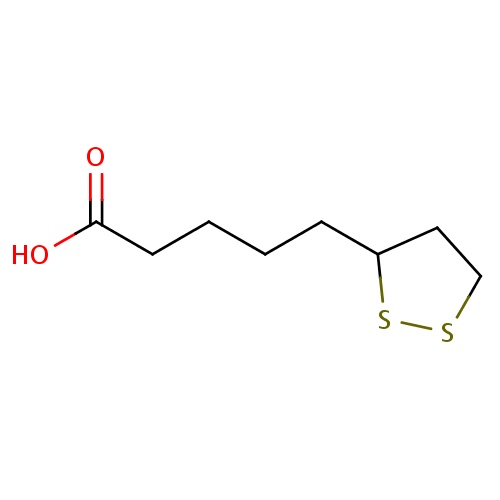
|
Vitamin B Complex; Antioxidants; Dietary Supplements; Micronutrients; Supplements; Alimentary Tract and Metabolism; Various Alimentary Tract and Metabolism Products; | For nutritional supplementation, also for treating dietary shortage or imbalance. |
| FDBD00199 | Valproic Acid |
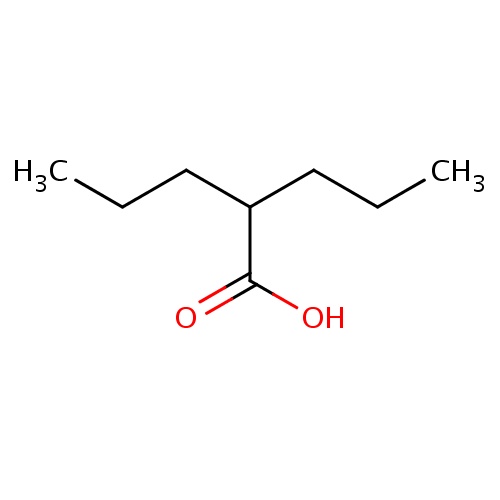
|
Enzyme Inhibitors; Anticonvulsants; Antimanic Agents; GABA Agents; Nervous System; Antiepileptics; Fatty Acid Derivatives; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2A6 Inhibitors; CYP2A6 Inhibitors (strong); CYP2A6 Inhibitors (moderate); CYP2A6 Inducers; CYP2A6 Inducers (strong); CYP2B6 Inhibitors (strong); CYP3A4 Inhibitors; | For treatment and management of seizure disorders, mania, and prophylactic treatment of migraine headache. In epileptics, valproic acid is used to control absence seizures, tonic-clonic seizures (grand mal), complex partial seizures, and the seizures associated with Lennox-Gastaut syndrome. |
| FDBD00289 | Mupirocin |
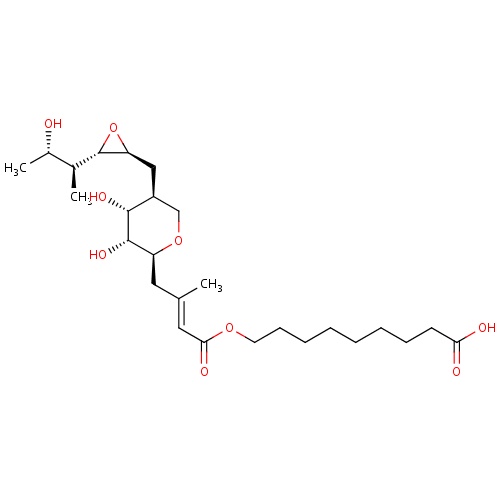
|
Anti-Bacterial Agents; Protein Synthesis Inhibitors; Antibiotics; Respiratory System; Dermatologicals; Nasal Preparations; Antibiotics for Topical Use; | For the treatment of . |
| FDBD00383 | Aminocaproic Acid |
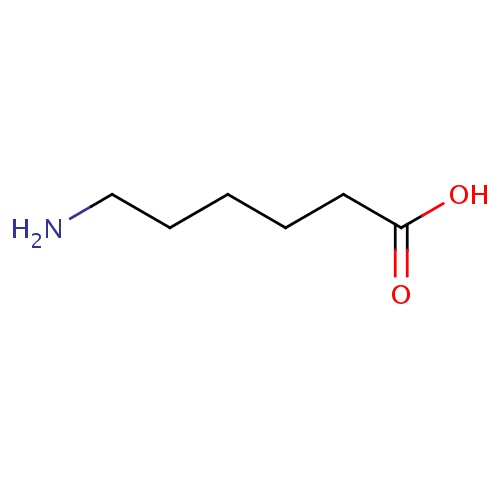
|
Antifibrinolytic Agents; Blood and Blood Forming Organs; Antihemorrhagics; Amino Acids; | For use in the treatment of excessive postoperative bleeding. |
| FDBD00387 | Anisotropine Methylbromide |
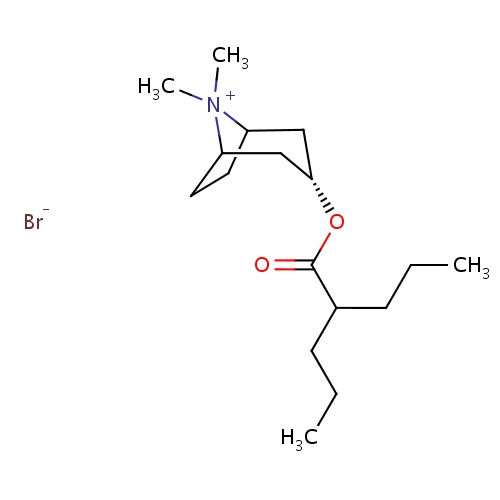
|
Muscarinic Antagonists; Cholinergic Antagonists; | For use in conjunction with antacids or histamine H. |
| FDBD00415 | Azelaic Acid |
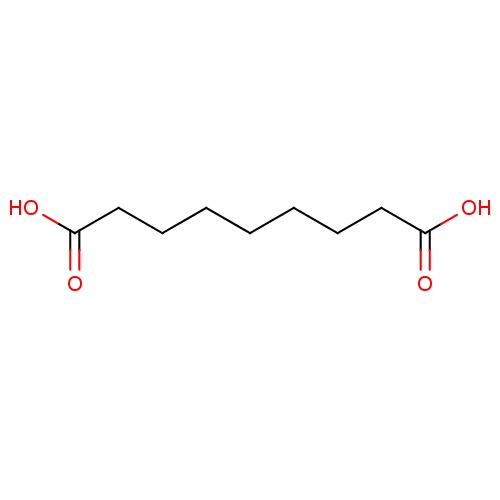
|
Antineoplastic Agents; Dermatologic Agents; Anti-Acne Preparations; Dermatologicals; Anti-Acne Preparations for Topical Use; | For the topical treatment of mild-to-moderate inflammatory acne vulgaris. |
| FDBD00633 | Alprostadil |
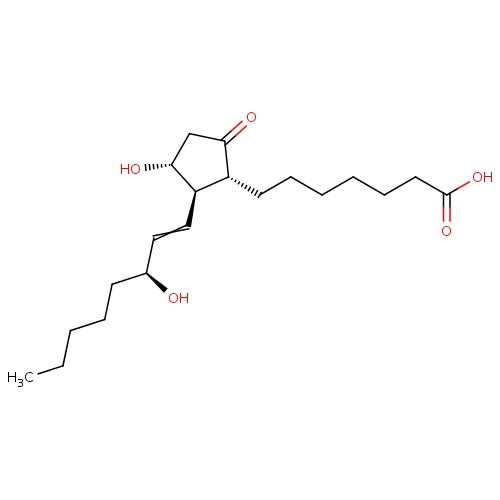
|
Platelet Aggregation Inhibitors; Vasodilator Agents; Prostaglandins; Genito Urinary System and Sex Hormones; Cardiovascular System; Drugs Used in Erectile Dysfunction; Cardiac Therapy; Urological Agents; | For palliative, not definitive, therapy to temporarily maintain the patency of the ductus arteriosus until corrective or palliative surgery can be performed in neonates who have congenital heart defects and who depend upon the patent ductus for survival. Also for the treatment of erectile dysfunction due to neurogenic, vasculogenic, psychogenic, or mixed etiology. |
23 ,
3
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 2c1q_ligand_3_0.mol2 | 2c1q | 1 | -6.73 | CCCCC(=O)O | 7 |
| 4qfo_ligand_2_20.mol2 | 4qfo | 1 | -6.72 | C(C(=O)O)CC(C)C | 8 |
| 2avi_ligand_3_0.mol2 | 2avi | 1 | -6.64 | C(=O)(O)CCCC | 7 |
| 4pox_ligand_2_12.mol2 | 4pox | 1 | -6.64 | C(C(=O)O)CCC | 7 |
| 4pow_ligand_3_3.mol2 | 4pow | 1 | -6.58 | C(C)CCC(=O)O | 7 |
| 2uyw_ligand_3_0.mol2 | 2uyw | 1 | -6.54 | CCCCC(=O)O | 7 |
| 4bj8_ligand_3_0.mol2 | 4bj8 | 1 | -6.54 | C(=O)(O)CCCC | 7 |
255 ,
26

