
IUPAC name
4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzene-1-sulfonamide
SMILES
CC1=CC=C(C=C1)C1=CC(=NN1C1=CC=C(C=C1)S(N)(=O)=O)C(F)(F)F
Compound class
Antineoplastic Agents; Cyclooxygenase 2 Inhibitors; Antineoplastic and Immunomodulating Agents; Musculo-Skeletal System; Antiinflammatory and Antirheumatic Products, Non-Steroids; Antiinflammatory and Antirheumatic Products; Coxibs; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors;
Therapeutic area
For relief and management of osteoarthritis (OA), rheumatoid arthritis (RA), juvenile rheumatoid arthritis (JRA), ankylosing spondylitis, acute pain, primary dysmenorrhea and oral adjunct to usual care for patients with familial adenomatous polyposis.
Common name
Celecoxib
IUPAC name
4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzene-1-sulfonamide
SMILES
CC1=CC=C(C=C1)C1=CC(=NN1C1=CC=C(C=C1)S(N)(=O)=O)C(F)(F)F
INCHI
InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25)
FORMULA
C17H14F3N3O2S

Common name
Celecoxib
IUPAC name
4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzene-1-sulfonamide
Molecular weight
381.372
clogP
2.632
clogS
-4.859
HBond Acceptor
3
HBond Donor
2
Total Polar Surface Area
77.98
Number of Rings
3
Rotatable Bond
4
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00005 | benzene |
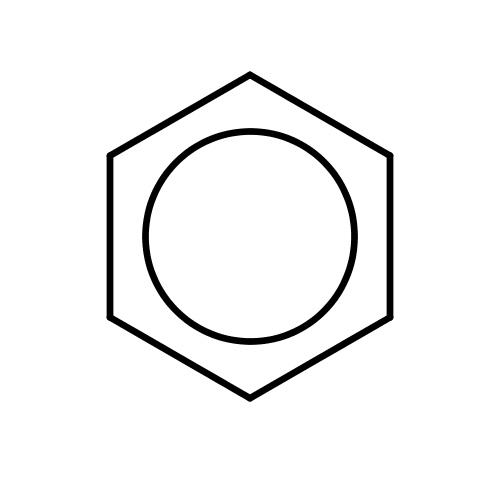
|
c1ccccc1 | 0.2824 |
| FDBF00023 | toluene |
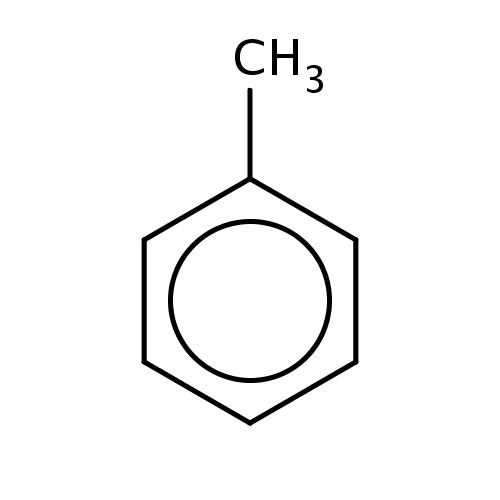
|
c1(ccccc1)C | 0.1268 |
| FDBF00177 | fluoroform |
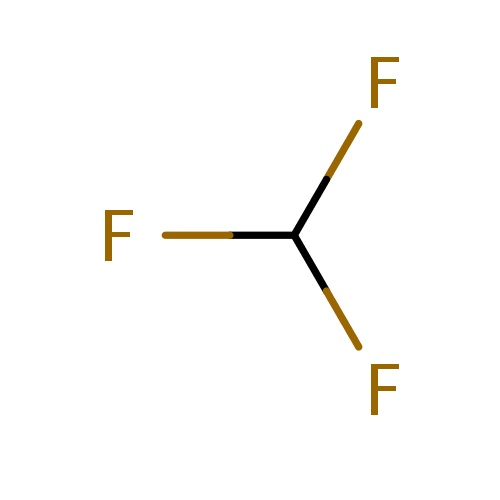
|
FC(F)F | 0.0704 |
| FDBF00567 | 1H-pyrazole |

|
[nH]1nccc1 | 0.0021 |
| FDBF01053 | 1-phenylpyrazole |
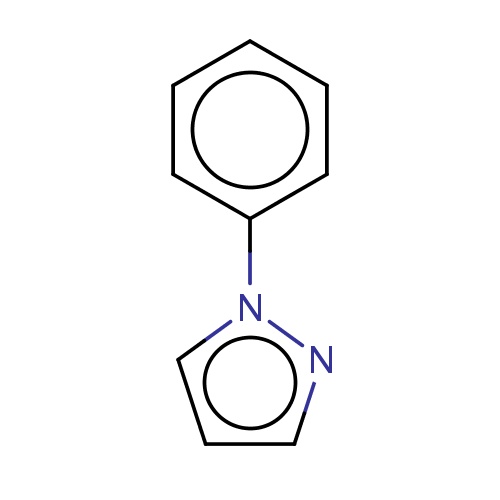
|
n1(nccc1)c2ccccc2 | 0.0007 |
| FDBF01054 | 3-(trifluoromethyl)-1H-pyrazole |
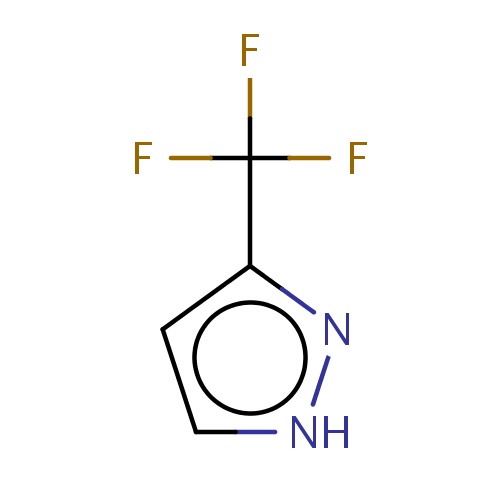
|
FC(F)(F)c1n[nH]cc1 | 0.0003 |
| FDBF01055 | 1-phenyl-5-(p-tolyl)pyrazole |
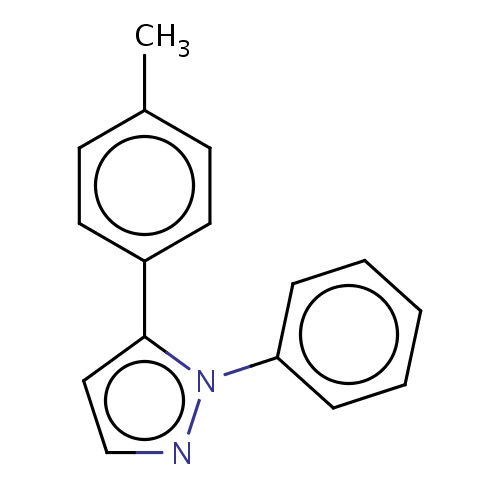
|
c1(ccc(cc1)C)c2n(ncc2)c3ccccc3 | 0.0003 |
| FDBF01056 | 1-phenyl-3-(trifluoromethyl)pyrazole |
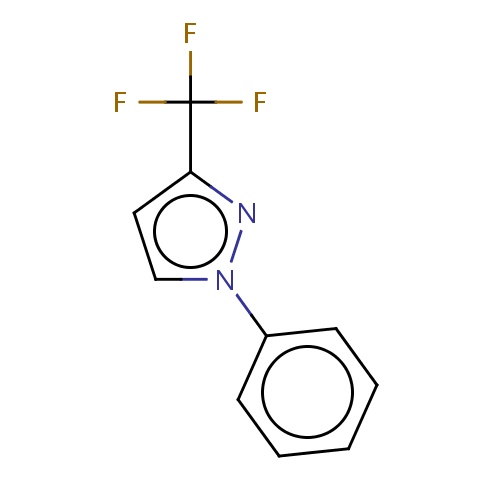
|
FC(F)(F)c1nn(cc1)c2ccccc2 | 0.0003 |

