
IUPAC name
5-methyl-2-[(5-methyl-1H-imidazol-4-yl)methyl]-1H,2H,3H,4H,5H-pyrido[4,3-b]indol-1-one
SMILES
CN1C2=C(C3=CC=CC=C13)C(=O)N(CC1=C(C)NC=N1)CC2
Compound class
Gastrointestinal Agents; Serotonin Antagonists; Alimentary Tract and Metabolism; Drugs for Functional Gastrointestinal Disorders; Serotonin Receptor Antagonists; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; Antiemetics Antagonists;
Therapeutic area
Only for the treatment of symptoms of severe diarrhea-predominant irritable bowel syndrome (IBS) in women with chronic symptoms (generally lasting greater than 6 months) who does not present with anatomic or biochemical GI abnormalities and have not responded to conventional therapy.
Common name
Alosetron
IUPAC name
5-methyl-2-[(5-methyl-1H-imidazol-4-yl)methyl]-1H,2H,3H,4H,5H-pyrido[4,3-b]indol-1-one
SMILES
CN1C2=C(C3=CC=CC=C13)C(=O)N(CC1=C(C)NC=N1)CC2
INCHI
InChI=1S/C17H18N4O/c1-11-13(19-10-18-11)9-21-8-7-15-16(17(21)22)12-5-3-4-6-14(12)20(15)2/h3-6,10H,7-9H2,1-2H3,(H,18,19)
FORMULA
C17H18N4O

Common name
Alosetron
IUPAC name
5-methyl-2-[(5-methyl-1H-imidazol-4-yl)methyl]-1H,2H,3H,4H,5H-pyrido[4,3-b]indol-1-one
Molecular weight
294.351
clogP
2.879
clogS
-4.282
HBond Acceptor
2
HBond Donor
1
Total Polar Surface Area
53.92
Number of Rings
4
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00051 | 5-methyl-1H-imidazole |
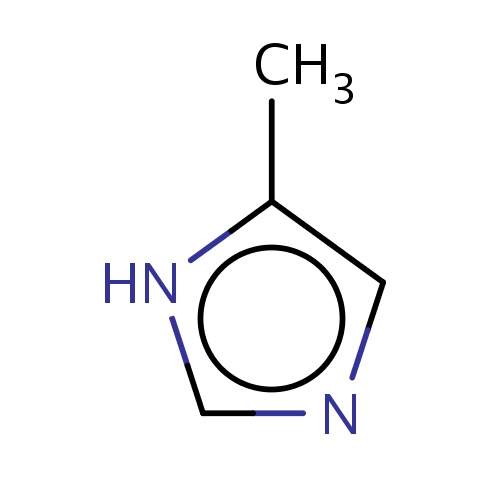
|
[nH]1c(cnc1)C | 0.0021 |
| FDBF01113 | 4,5-dimethyl-1H-imidazole |
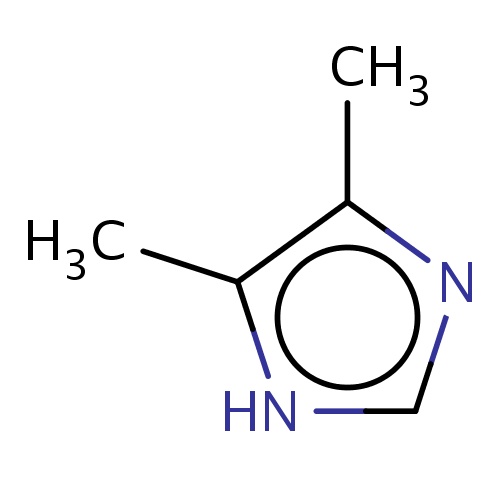
|
[nH]1c(c(nc1)C)C | 0.0007 |
| FDBF02190 | 2,5-dimethyl-3,4-dihydropyrido[4,3-b]indol-1-one |
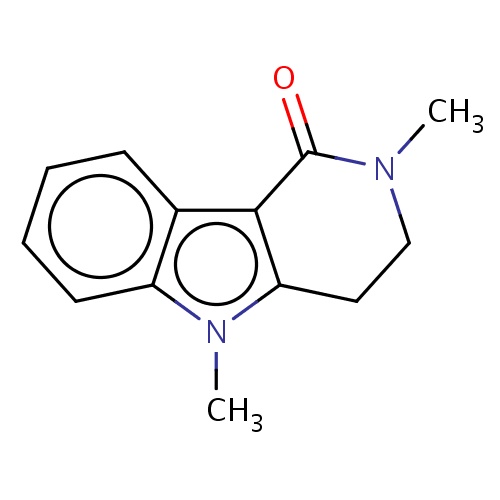
|
O=C1N(CCc2c1c3c(n2C)cccc3)C | 0.0003 |
| FDBF02191 | 5-methyl-3,4-dihydro-2H-pyrido[4,3-b]indol-1-one |
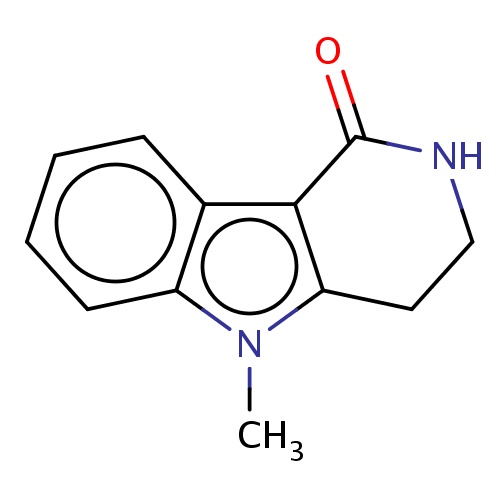
|
O=C1NCCc2c1c3c(n2C)cccc3 | 0.0003 |

