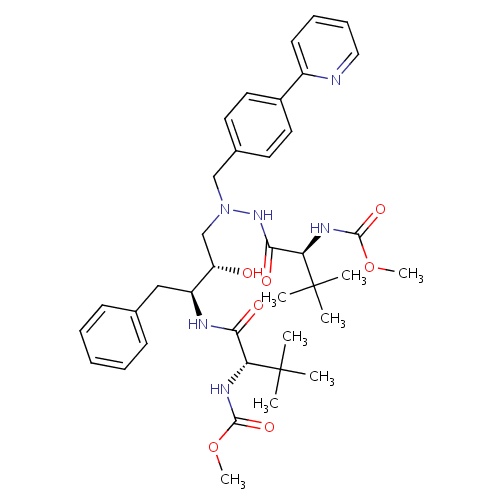
IUPAC name
methyl N-[(1S)-1-{[(2S,3S)-3-hydroxy-4-[(2S)-2-[(methoxycarbonyl)amino]-3,3-dimethyl-N'-{[4-(pyridin-2-yl)phenyl]methyl}butanehydrazido]-1-phenylbutan-2-yl]carbamoyl}-2,2-dimethylpropyl]carbamate
SMILES
COC(=O)N[C@H](C(=O)N[C@@H](CC1=CC=CC=C1)[C@@H](O)CN(CC1=CC=C(C=C1)C1=CC=CC=N1)NC(=O)[C@@H](NC(=O)OC)C(C)(C)C)C(C)(C)C
Compound class
Anti-HIV Agents; Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein;
Therapeutic area
Used in combination with other antiretroviral agents for the treatment of HIV-1 infection, as well as postexposure prophylaxis of HIV infection in individuals who have had occupational or nonoccupational exposure to potentially infectious body fluids of a person known to be infected with HIV when that exposure represents a substantial risk for HIV transmission.
Common name
Atazanavir
IUPAC name
methyl N-[(1S)-1-{[(2S,3S)-3-hydroxy-4-[(2S)-2-[(methoxycarbonyl)amino]-3,3-dimethyl-N'-{[4-(pyridin-2-yl)phenyl]methyl}butanehydrazido]-1-phenylbutan-2-yl]carbamoyl}-2,2-dimethylpropyl]carbamate
SMILES
COC(=O)N[C@H](C(=O)N[C@@H](CC1=CC=CC=C1)[C@@H](O)CN(CC1=CC=C(C=C1)C1=CC=CC=N1)NC(=O)[C@@H](NC(=O)OC)C(C)(C)C)C(C)(C)C
INCHI
InChI=1S/C38H52N6O7/c1-37(2,3)31(41-35(48)50-7)33(46)40-29(22-25-14-10-9-11-15-25)30(45)24-44(43-34(47)32(38(4,5)6)42-36(49)51-8)23-26-17-19-27(20-18-26)28-16-12-13-21-39-28/h9-21,29-32,45H,22-24H2,1-8H3,(H,40,46)(H,41,48)(H,42,49)(H,43,47)/t29-,30-,31+,32+/m0/s1
FORMULA
C38H52N6O7

Common name
Atazanavir
IUPAC name
methyl N-[(1S)-1-{[(2S,3S)-3-hydroxy-4-[(2S)-2-[(methoxycarbonyl)amino]-3,3-dimethyl-N'-{[4-(pyridin-2-yl)phenyl]methyl}butanehydrazido]-1-phenylbutan-2-yl]carbamoyl}-2,2-dimethylpropyl]carbamate
Molecular weight
704.855
clogP
4.112
clogS
-7.488
HBond Acceptor
9
HBond Donor
5
Total Polar Surface Area
171.22
Number of Rings
3
Rotatable Bond
18
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00014 | pyridine |
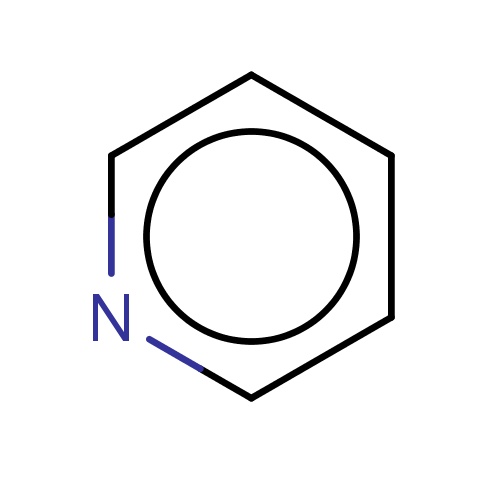
|
c1cccnc1 | 0.0333 |
| FDBF00015 | acetamide |
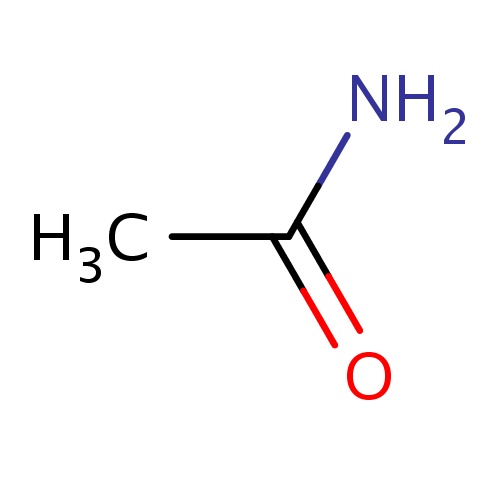
|
NC(=O)C | 0.0460 |
| FDBF00023 | toluene |
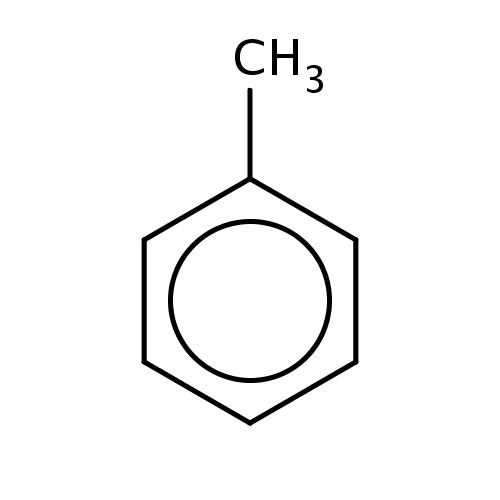
|
c1(ccccc1)C | 0.1268 |
| FDBF00043 | N-(2-hydroxyethyl)formamide |

|
C(CO)NC=O | 0.0065 |
| FDBF00066 | N-methylmethanamine |
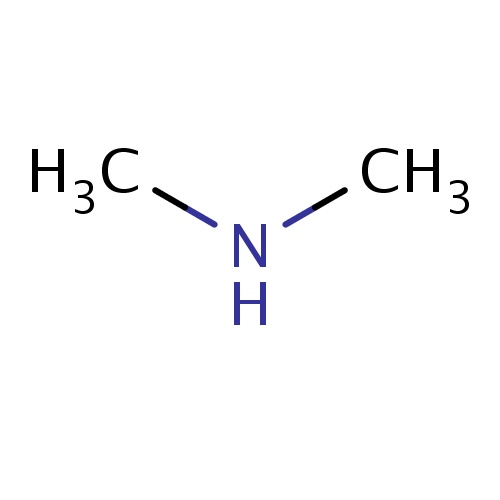
|
N(C)C | 0.0914 |
| FDBF00143 | N-phenethylformamide |

|
c1(ccccc1)CCNC=O | 0.0041 |
| FDBF01780 | N,3,3-trimethylbutanamide |

|
O=C(NC)CC(C)(C)C | 0.0007 |
| FDBF02376 | [4-(2-pyridyl)phenyl]methanamine |

|
n1c(cccc1)c2ccc(cc2)CN | 0.0003 |
| FDBF02393 | N',N',3,3-tetramethylbutanehydrazide |
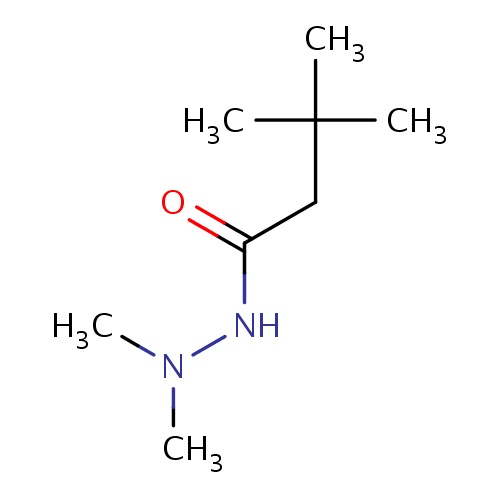
|
CN(C)NC(=O)CC(C)(C)C | 0.0003 |

