
Common name
N-phenethylformamide
IUPAC name
N-phenethylformamide
SMILES
c1(ccccc1)CCNC=O
Common name
N-phenethylformamide
IUPAC name
N-phenethylformamide
SMILES
c1(ccccc1)CCNC=O
INCHI
InChI=1S/C9H11NO/c11-8-10-7-6-9-4-2-1-3-5-9/h1-5,8H,6-7H2,(H,10,11)
FORMULA
C9H11NO

Common name
N-phenethylformamide
IUPAC name
N-phenethylformamide
Molecular weight
149.190
clogP
1.862
clogS
-2.431
Frequency
0.0041
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
29.1
Number of Rings
1
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00057 | Aspartame |
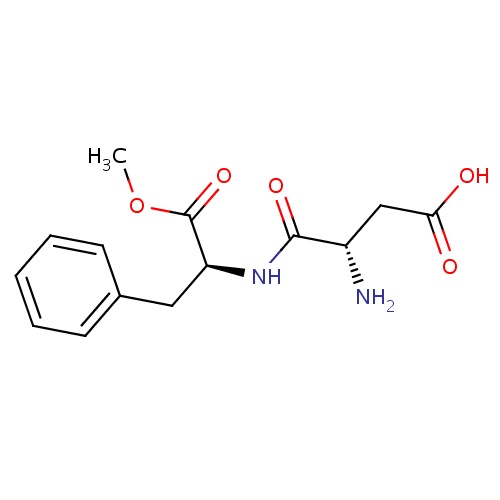
|
Sweetening Agents; Dietary Supplements; Micronutrients; Supplements; | Used as a diet supplement and sugar substitute. |
| FDBD00077 | Bortezomib |
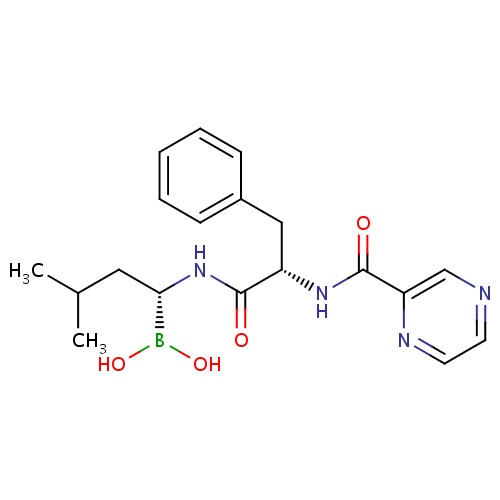
|
Antineoplastic Agents; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For treatment of multiple myeloma in patients who have not been successfully treated with at least two previous therapies. |
| FDBD00376 | Ritonavir |

|
Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; Combined Inducers of CYP3A4 and P-glycoprotein; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Indicated in combination with other antiretroviral agents for the treatment of HIV-infection. |
| FDBD00870 | Glyburide |
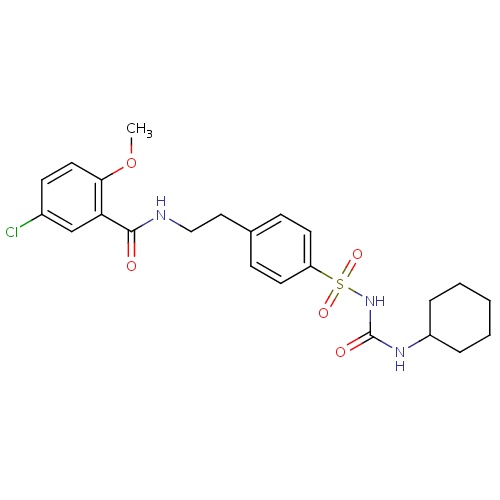
|
Hypoglycemic Agents; Anti-Arrhythmia Agents; Drugs Used in Diabetes; Alimentary Tract and Metabolism; Blood Glucose Lowering Drugs, Excl. Insulins; Sulfonylureas; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP3A4 Inhibitors; BSEP/ABCB11 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Indicated as an adjunct to diet to lower the blood glucose in patients with NIDDM whose hyperglycemia cannot be satisfactorily controlled by diet alone. |
| FDBD00918 | Glipizide |
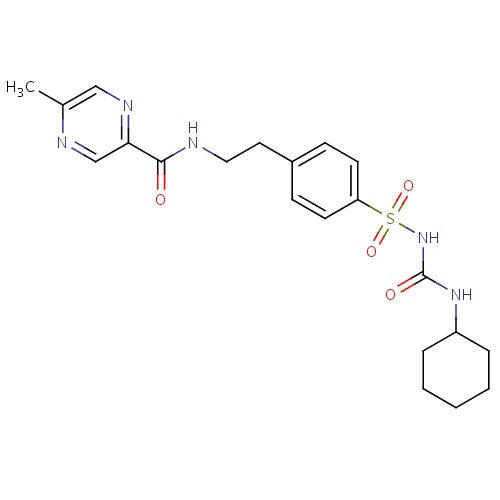
|
Hypoglycemic Agents; Drugs Used in Diabetes; Alimentary Tract and Metabolism; Blood Glucose Lowering Drugs, Excl. Insulins; Sulfonylureas; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; CYP3A4 Inhibitors; | For use as an adjunct to diet for the control of hyperglycemia and its associated symptomatology in patients with non-insulin-dependent diabetes mellitus (NIDDM; type II), formerly known as maturity-onset diabetes, after an adequate trial of dietary therapy has proved unsatisfactory. |
| FDBD00923 | Atazanavir |
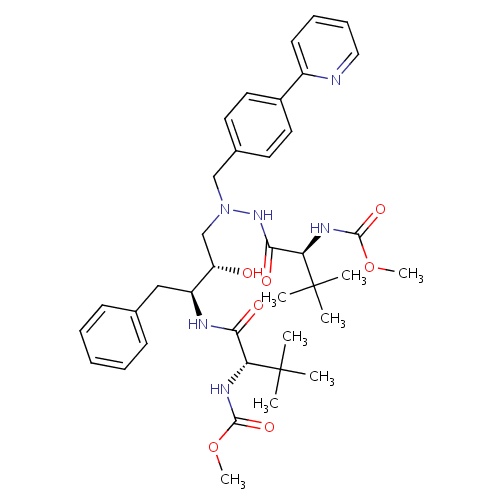
|
Anti-HIV Agents; Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Used in combination with other antiretroviral agents for the treatment of HIV-1 infection, as well as postexposure prophylaxis of HIV infection in individuals who have had occupational or nonoccupational exposure to potentially infectious body fluids of a person known to be infected with HIV when that exposure represents a substantial risk for HIV transmission. |
| FDBD01076 | Saquinavir |
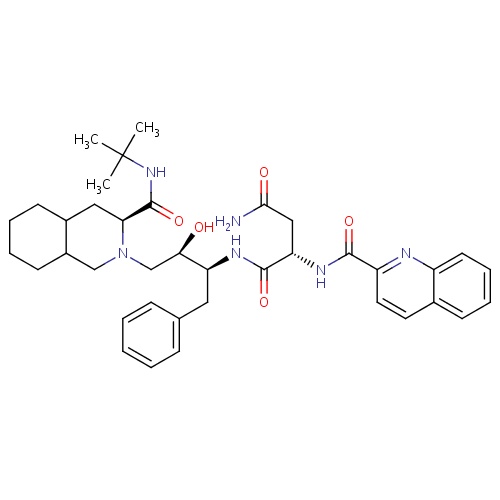
|
Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | For the treatment of HIV-1 with advanced immunodeficiency together with antiretroviral nucleoside analogues. |
| FDBD01117 | Glisoxepide |
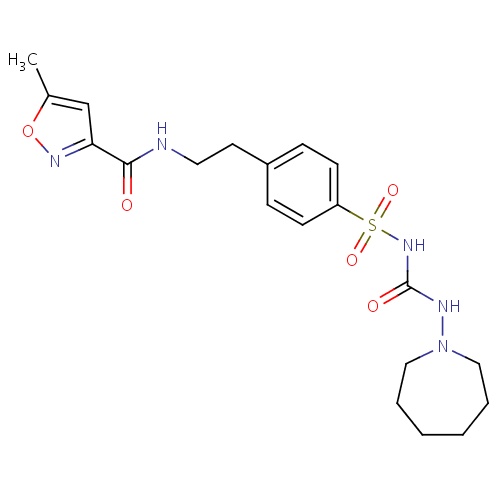
|
Drugs Used in Diabetes; Alimentary Tract and Metabolism; Blood Glucose Lowering Drugs, Excl. Insulins; Sulfonylureas; | For the treatment of diabetes mellitus type 2. |
| FDBD01173 | Bezafibrate |

|
Hypolipidemic Agents; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Fibrates; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; | For the treatment of primary hyperlipidaemia types IIa, IIb, III, IV and V (Fredrickson classification) corresponding to groups I, II and III of the European Atherosclerosis Society guidelines - when diet alone or improvements in lifestyle such as increased exercise or weight reduction do not lead to an adequate response. Also for the treatment of secondary hyperlipidaemias, e.g. severe hypertriglyceridemias, when sufficient improvement does not occur after correction of the underlying disorder (e.g. diabetes mellitus). |
| FDBD01251 | Lopinavir |
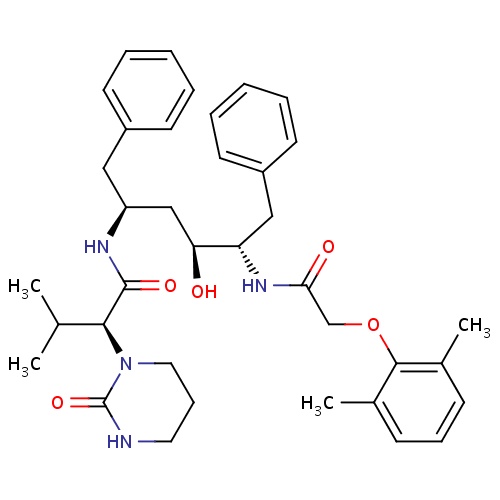
|
Anti-HIV Agents; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP3A Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Indicated in combination with other antiretroviral agents for the treatment of HIV-infection. |
12 ,
2
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1mj7_ligand_3_0.mol2 | 1mj7 | 1 | -7.34 | N(C=O)CCc1ccccc1 | 11 |
| 2xfk_ligand_3_203.mol2 | 2xfk | 1 | -7.32 | C(=O)NCCc1ccccc1 | 11 |
| 2xfi_ligand_3_130.mol2 | 2xfi | 1 | -7.27 | c1(ccccc1)CCNC=O | 11 |
| 2viz_ligand_3_167.mol2 | 2viz | 1 | -7.25 | C(=O)NCCc1ccccc1 | 11 |
| 2vj9_ligand_3_86.mol2 | 2vj9 | 1 | -7.24 | C(CNC=O)c1ccccc1 | 11 |
| 1qf1_ligand_3_113.mol2 | 1qf1 | 1 | -7.23 | c1(ccccc1)CCNC=O | 11 |
| 2vj6_ligand_3_137.mol2 | 2vj6 | 1 | -7.23 | C(=O)NCCc1ccccc1 | 11 |
| 2xfj_ligand_3_137.mol2 | 2xfj | 1 | -7.23 | C(Cc1ccccc1)NC=O | 11 |
| 2viy_ligand_3_90.mol2 | 2viy | 1 | -7.22 | c1(ccccc1)CCNC=O | 11 |
| 2vkm_ligand_3_231.mol2 | 2vkm | 1 | -7.22 | C(=O)NCCc1ccccc1 | 11 |
292 ,
30

