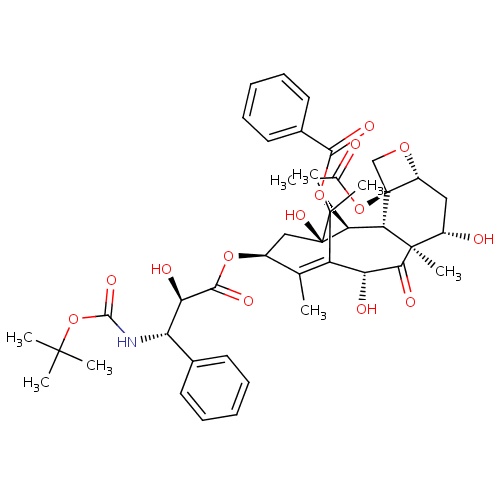
IUPAC name
(1S,2S,3R,4S,7R,9S,10S,12R,15S)-4-(acetyloxy)-15-{[(2R,3S)-3-{[(tert-butoxy)carbonyl]amino}-2-hydroxy-3-phenylpropanoyl]oxy}-1,9,12-trihydroxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.0³,¹
SMILES
[H][C@@]12C[C@H](O)[C@@]3(C)C(=O)[C@H](O)C4=C(C)[C@H](C[C@@](O)([C@@H](OC(=O)C5=CC=CC=C5)[C@]3([H])[C@@]1(CO2)OC(C)=O)C4(C)C)OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C1=CC=CC=C1
Compound class
Antineoplastic Agents; Immunosuppressive Agents; Tubulin Modulators; Antineoplastic and Immunomodulating Agents; Taxanes; CYP3A4 Inhibitors;
Therapeutic area
For the treatment of patients with locally advanced or metastatic breast cancer after failure of prior chemotherapy. Also used as a single agent in the treatment of patients with locally advanced or metastatic non-small cell lung cancer after failure of prior platinum-based chemotherapy. It is also used in combination with prednisone, in the treatment of patients with androgen independent (hormone refractory) metastatic prostate cancer. Furthermore, docetaxel has uses in the treatment of gastric adenocarinoma and head and neck cancer.
Common name
Docetaxel
IUPAC name
(1S,2S,3R,4S,7R,9S,10S,12R,15S)-4-(acetyloxy)-15-{[(2R,3S)-3-{[(tert-butoxy)carbonyl]amino}-2-hydroxy-3-phenylpropanoyl]oxy}-1,9,12-trihydroxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.0³,¹
SMILES
[H][C@@]12C[C@H](O)[C@@]3(C)C(=O)[C@H](O)C4=C(C)[C@H](C[C@@](O)([C@@H](OC(=O)C5=CC=CC=C5)[C@]3([H])[C@@]1(CO2)OC(C)=O)C4(C)C)OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C1=CC=CC=C1
INCHI
InChI=1S/C43H53NO14/c1-22-26(55-37(51)32(48)30(24-15-11-9-12-16-24)44-38(52)58-39(3,4)5)20-43(53)35(56-36(50)25-17-13-10-14-18-25)33-41(8,34(49)31(47)29(22)40(43,6)7)27(46)19-28-42(33,21-54-28)57-23(2)45/h9-18,26-28,30-33,35,46-48,53H,19-21H2,1-8H3,(H,44,52)/t26-,27-,28+,30-,31+,32+,33-,35-,41+,42-,43+/m0/s1
FORMULA
C43H53NO14

Common name
Docetaxel
IUPAC name
(1S,2S,3R,4S,7R,9S,10S,12R,15S)-4-(acetyloxy)-15-{[(2R,3S)-3-{[(tert-butoxy)carbonyl]amino}-2-hydroxy-3-phenylpropanoyl]oxy}-1,9,12-trihydroxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.0³,¹
Molecular weight
807.879
clogP
3.512
clogS
-5.148
HBond Acceptor
14
HBond Donor
5
Total Polar Surface Area
224.45
Number of Rings
6
Rotatable Bond
13
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
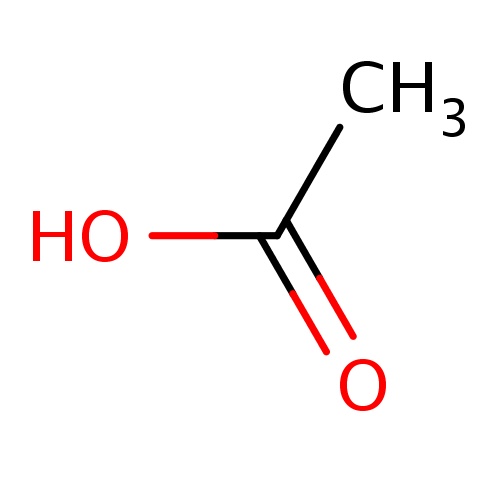
| FDBF00004 | acetic acid |
|
CC(=O)O | 0.0687 |
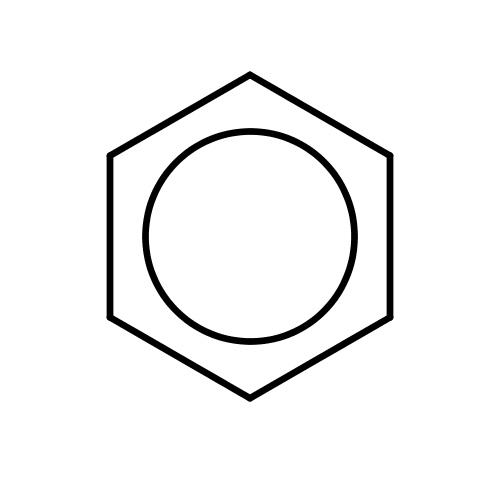
| FDBF00005 | benzene |
|
c1ccccc1 | 0.2824 |
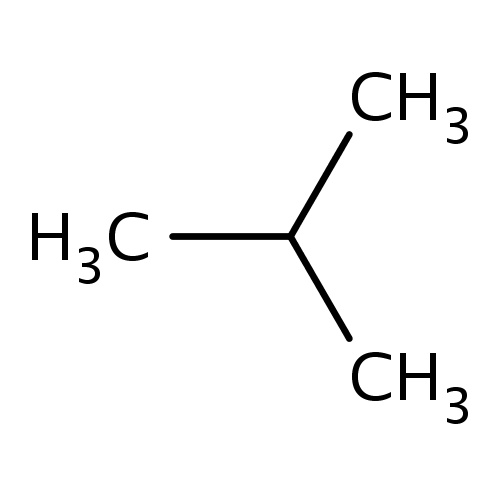
| FDBF00012 | isobutane |
|
C(C)(C)C | 0.0611 |
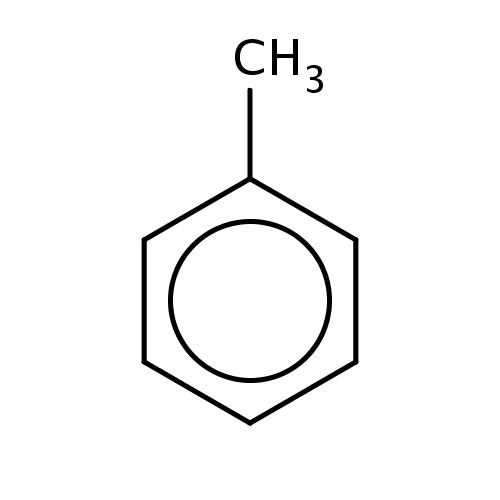
| FDBF00023 | toluene |
|
c1(ccccc1)C | 0.1268 |
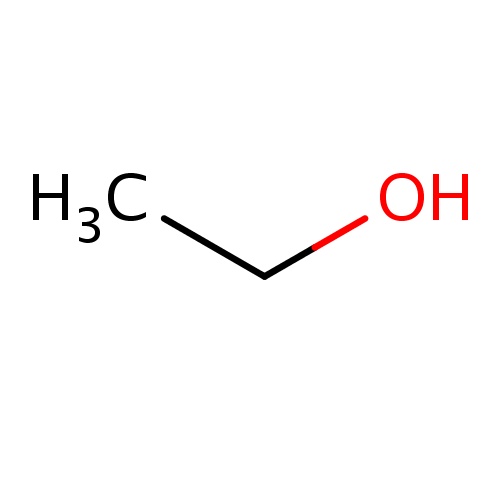
| FDBF00041 | ethanol |
|
CCO | 0.1474 |
| FDBF00253 | 2-phenylethanol |

|
c1(ccccc1)CCO | 0.0038 |
| FDBF01127 | methylcarbamic acid |

|
O=C(O)NC | 0.0199 |
| FDBF02839 | tert-butyl carbamate |

|
O(C(=O)N)C(C)(C)C | 0.0010 |
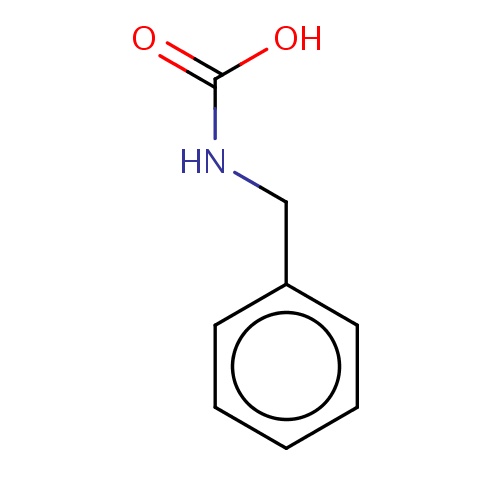
| FDBF02840 | benzylcarbamic acid |
|
O=C(O)NCc1ccccc1 | 0.0003 |
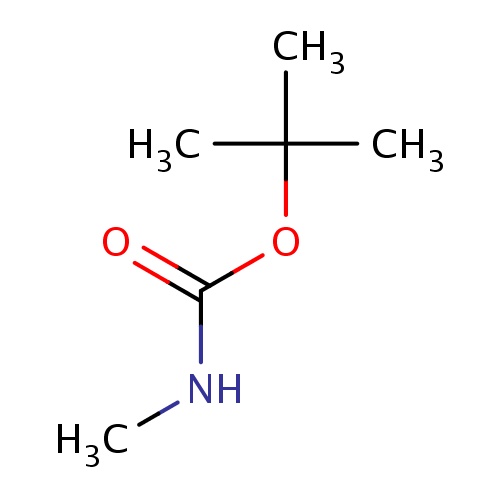
| FDBF02841 | tert-butyl N-methylcarbamate |
|
O(C(=O)NC)C(C)(C)C | 0.0003 |

