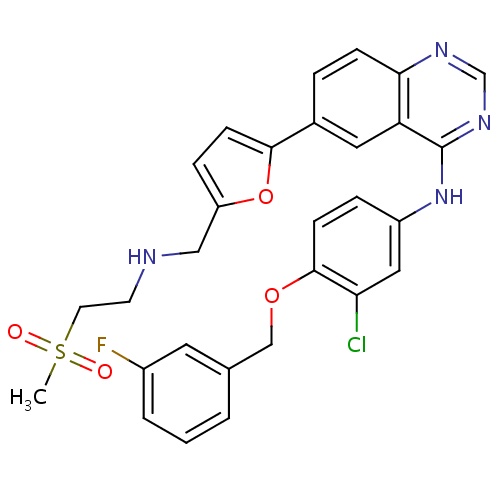
IUPAC name
N-{3-chloro-4-[(3-fluorophenyl)methoxy]phenyl}-6-(5-{[(2-methanesulfonylethyl)amino]methyl}furan-2-yl)quinazolin-4-amine
SMILES
CS(=O)(=O)CCNCC1=CC=C(O1)C1=CC2=C(C=C1)N=CN=C2NC1=CC(Cl)=C(OCC2=CC(F)=CC=C2)C=C1
Compound class
Antineoplastic Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein;
Therapeutic area
Indicated in combination with capecitabine for the treatment of patients with advanced or metastatic breast cancer whose tumors overexpress the human epidermal receptor type 2 (HER2) protein and who have received prior therapy including an anthracycline, a taxane, and trastuzuma.
Common name
Lapatinib
IUPAC name
N-{3-chloro-4-[(3-fluorophenyl)methoxy]phenyl}-6-(5-{[(2-methanesulfonylethyl)amino]methyl}furan-2-yl)quinazolin-4-amine
SMILES
CS(=O)(=O)CCNCC1=CC=C(O1)C1=CC2=C(C=C1)N=CN=C2NC1=CC(Cl)=C(OCC2=CC(F)=CC=C2)C=C1
INCHI
InChI=1S/C29H26ClFN4O4S/c1-40(36,37)12-11-32-16-23-7-10-27(39-23)20-5-8-26-24(14-20)29(34-18-33-26)35-22-6-9-28(25(30)15-22)38-17-19-3-2-4-21(31)13-19/h2-10,13-15,18,32H,11-12,16-17H2,1H3,(H,33,34,35)
FORMULA
C29H26ClFN4O4S

Common name
Lapatinib
IUPAC name
N-{3-chloro-4-[(3-fluorophenyl)methoxy]phenyl}-6-(5-{[(2-methanesulfonylethyl)amino]methyl}furan-2-yl)quinazolin-4-amine
Molecular weight
581.058
clogP
5.845
clogS
-10.519
HBond Acceptor
6
HBond Donor
2
Total Polar Surface Area
106.35
Number of Rings
5
Rotatable Bond
11
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
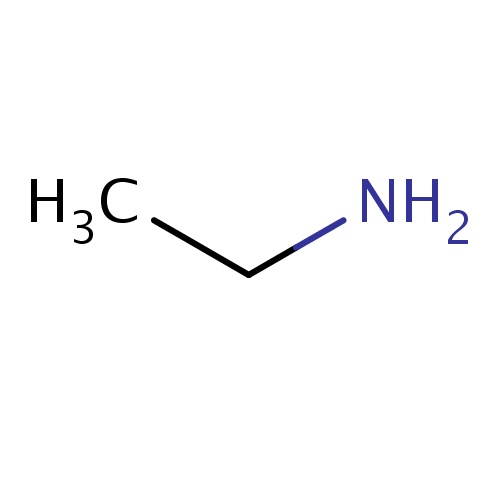
| FDBF00040 | ethanamine |
|
CCN | 0.0677 |
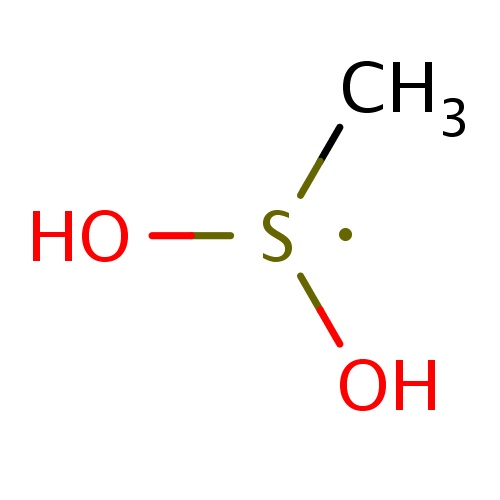
| FDBF00317 | (dihydroxy-λ3-sulfanyl)methane |
|
C[S](O)O | 0.0141 |
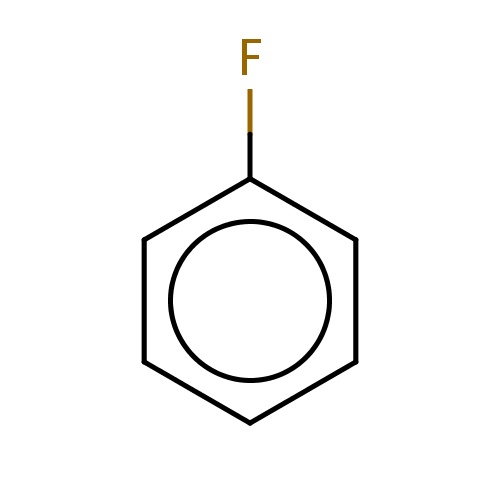
| FDBF00378 | fluorobenzene |
|
Fc1ccccc1 | 0.0237 |
| FDBF01001 | furan |
|
o1cccc1 | 0.0079 |
| FDBF01618 | 2-methylfuran |
|
Cc1occc1 | 0.0055 |
| FDBF02019 | 1-methylsulfonylethane |

|
CS(=O)(=O)CC | 0.0017 |
| FDBF02629 | 3-chloroaniline |

|
Nc1cccc(c1)Cl | 0.0010 |
| FDBF02864 | methylsulfonylmethane |

|
S(=O)(=O)(C)C | 0.0010 |
| FDBF02866 | 2-chlorophenol |

|
Clc1ccccc1O | 0.0055 |
| FDBF02873 | 2-chloro-4-(quinazolin-4-ylamino)phenol |
|
Clc1cc(ccc1O)Nc2c3c(ncn2)cccc3 | 0.0003 |

