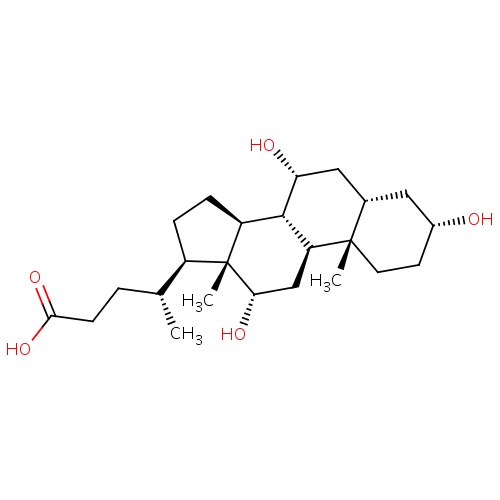
IUPAC name
(4R)-4-[(1S,2S,5R,7S,9R,10R,11S,14R,15R,16S)-5,9,16-trihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,
SMILES
[H][C@@](C)(CCC(O)=O)[C@@]1([H])CC[C@@]2([H])[C@]3([H])[C@]([H])(O)C[C@]4([H])C[C@]([H])(O)CC[C@]4(C)[C@@]3([H])C[C@]([H])(O)[C@]12C
Compound class
Alimentary Tract and Metabolism; Bile and Liver Therapy; Bile Acid Preparations; Bile Therapy; BSEP/ABCB11 Inhibitors;
Therapeutic area
Oral cholic acid is indicated for: treatment of bile acid synthesis disorders due to single enzyme defects; and as adjunctive treatment of peroxisomal disorders including Zellweger spectrum disorders in patients who exhibit manifestations of liver disease, steatorrhea or complications from decreased fat soluble vitamin absorption.
Common name
Cholic Acid
IUPAC name
(4R)-4-[(1S,2S,5R,7S,9R,10R,11S,14R,15R,16S)-5,9,16-trihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,
SMILES
[H][C@@](C)(CCC(O)=O)[C@@]1([H])CC[C@@]2([H])[C@]3([H])[C@]([H])(O)C[C@]4([H])C[C@]([H])(O)CC[C@]4(C)[C@@]3([H])C[C@]([H])(O)[C@]12C
INCHI
InChI=1S/C24H40O5/c1-13(4-7-21(28)29)16-5-6-17-22-18(12-20(27)24(16,17)3)23(2)9-8-15(25)10-14(23)11-19(22)26/h13-20,22,25-27H,4-12H2,1-3H3,(H,28,29)/t13-,14+,15-,16-,17+,18+,19-,20+,22+,23+,24-/m1/s1
FORMULA
C24H40O5

Common name
Cholic Acid
IUPAC name
(4R)-4-[(1S,2S,5R,7S,9R,10R,11S,14R,15R,16S)-5,9,16-trihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,
Molecular weight
408.571
clogP
2.527
clogS
-2.311
HBond Acceptor
5
HBond Donor
4
Total Polar Surface Area
97.99
Number of Rings
4
Rotatable Bond
4