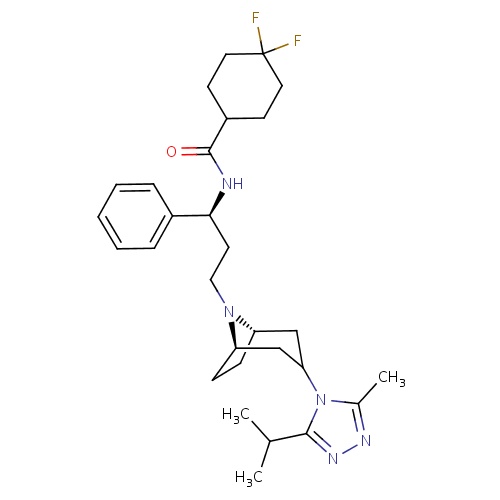
IUPAC name
4,4-difluoro-N-[(1S)-3-[(1R,5S)-3-[3-methyl-5-(propan-2-yl)-4H-1,2,4-triazol-4-yl]-8-azabicyclo[3.2.1]octan-8-yl]-1-phenylpropyl]cyclohexane-1-carboxamide
SMILES
[H][C@]12CC[C@]([H])(CC(C1)N1C(C)=NN=C1C(C)C)N2CC[C@H](NC(=O)C1CCC(F)(F)CC1)C1=CC=CC=C1
Compound class
Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; CYP3A4 Inhibitors; CCR5 Receptor Antagonists;
Therapeutic area
For treatment-experienced adult patients infected with only CCR5-tropic HIV-1 detectable, who have evidence of viral replication and HIV-1 strains resistant to multiple antiretroviral agents.
Common name
Maraviroc
IUPAC name
4,4-difluoro-N-[(1S)-3-[(1R,5S)-3-[3-methyl-5-(propan-2-yl)-4H-1,2,4-triazol-4-yl]-8-azabicyclo[3.2.1]octan-8-yl]-1-phenylpropyl]cyclohexane-1-carboxamide
SMILES
[H][C@]12CC[C@]([H])(CC(C1)N1C(C)=NN=C1C(C)C)N2CC[C@H](NC(=O)C1CCC(F)(F)CC1)C1=CC=CC=C1
INCHI
InChI=1S/C29H41F2N5O/c1-19(2)27-34-33-20(3)36(27)25-17-23-9-10-24(18-25)35(23)16-13-26(21-7-5-4-6-8-21)32-28(37)22-11-14-29(30,31)15-12-22/h4-8,19,22-26H,9-18H2,1-3H3,(H,32,37)/t23-,24+,25?,26-/m0/s1
FORMULA
C29H41F2N5O

Common name
Maraviroc
IUPAC name
4,4-difluoro-N-[(1S)-3-[(1R,5S)-3-[3-methyl-5-(propan-2-yl)-4H-1,2,4-triazol-4-yl]-8-azabicyclo[3.2.1]octan-8-yl]-1-phenylpropyl]cyclohexane-1-carboxamide
Molecular weight
513.666
clogP
4.779
clogS
-6.240
HBond Acceptor
4
HBond Donor
1
Total Polar Surface Area
63.05
Number of Rings
5
Rotatable Bond
8
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00003 | formamide |
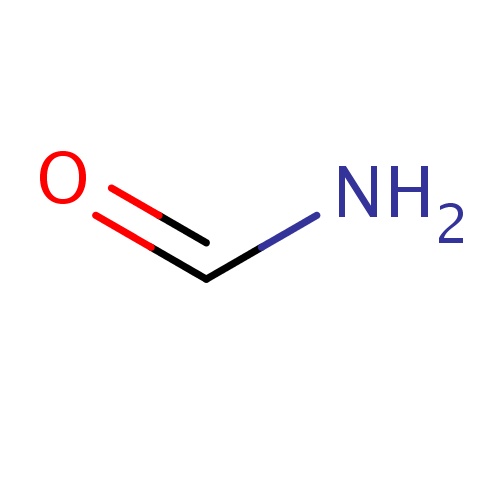
|
C(=O)N | 0.1240 |
| FDBF00182 | N-benzylformamide |
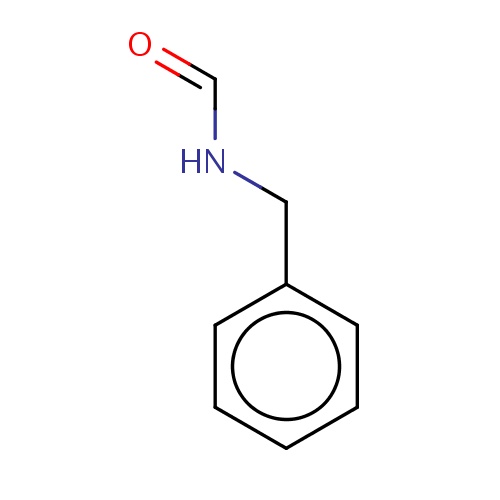
|
c1(ccccc1)CNC=O | 0.0038 |
| FDBF03161 | 4,4-difluorocyclohexanecarboxamide |
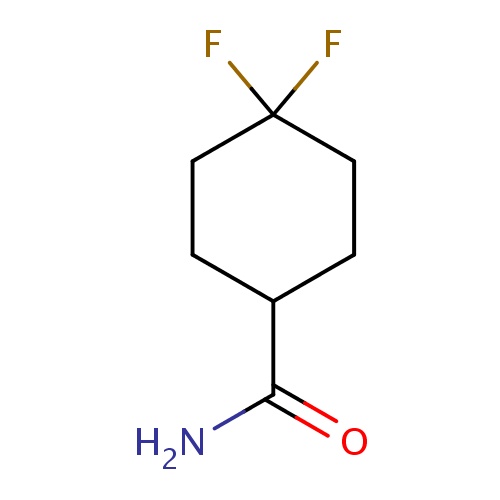
|
FC1(CCC(CC1)C(=O)N)F | 0.0003 |
| FDBF03169 | N-ethyl-4,4-difluoro-cyclohexanecarboxamide |
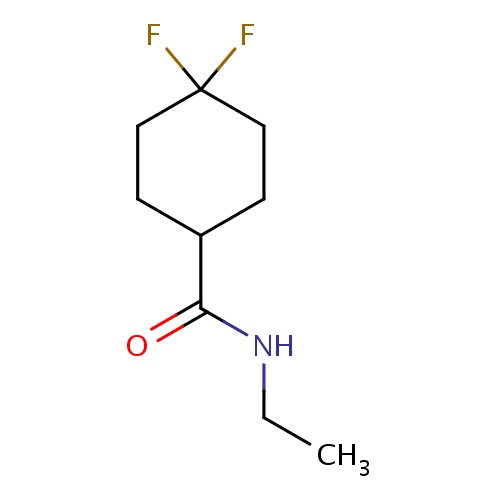
|
FC1(CCC(CC1)C(=O)NCC)F | 0.0003 |
| FDBF03174 | 4,4-difluoro-N-propyl-cyclohexanecarboxamide |
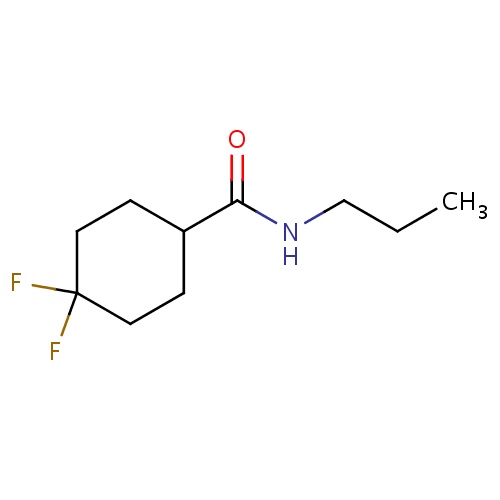
|
FC1(CCC(CC1)C(=O)NCCC)F | 0.0003 |
| FDBF03175 | 1,1-difluorocyclohexane |
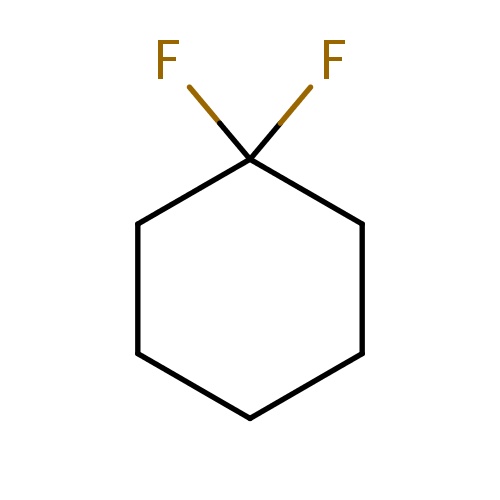
|
FC1(CCCCC1)F | 0.0003 |

