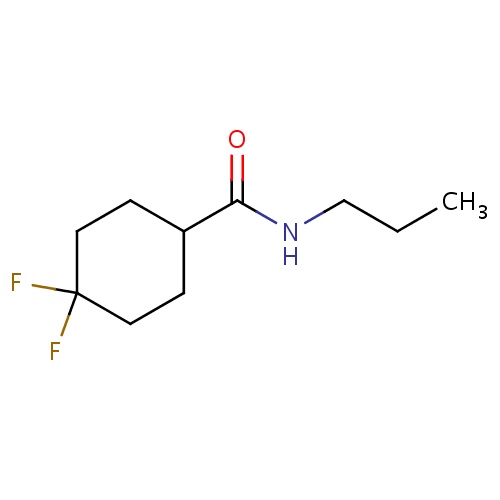
Common name
4,4-difluoro-N-propyl-cyclohexanecarboxamide
IUPAC name
4,4-difluoro-N-propyl-cyclohexanecarboxamide
SMILES
FC1(CCC(CC1)C(=O)NCCC)F
Common name
4,4-difluoro-N-propyl-cyclohexanecarboxamide
IUPAC name
4,4-difluoro-N-propyl-cyclohexanecarboxamide
SMILES
FC1(CCC(CC1)C(=O)NCCC)F
INCHI
InChI=1S/C10H17F2NO/c1-2-7-13-9(14)8-3-5-10(11,12)6-4-8/h8H,2-7H2,1H3,(H,13,14)
FORMULA
C10H17F2NO

Common name
4,4-difluoro-N-propyl-cyclohexanecarboxamide
IUPAC name
4,4-difluoro-N-propyl-cyclohexanecarboxamide
Molecular weight
205.245
clogP
2.588
clogS
-3.067
Frequency
0.0003
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
29.1
Number of Rings
1
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01321 | Maraviroc |
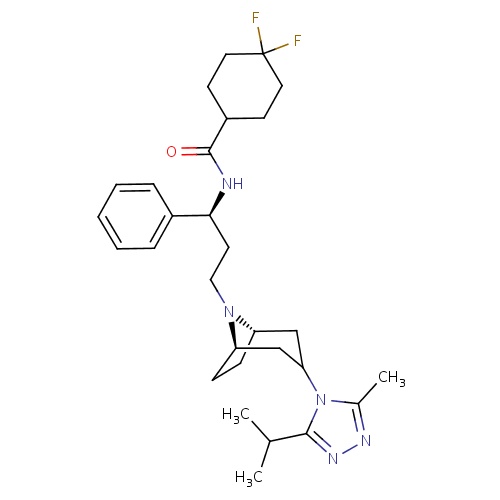
|
Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; CYP3A4 Inhibitors; CCR5 Receptor Antagonists; | For treatment-experienced adult patients infected with only CCR5-tropic HIV-1 detectable, who have evidence of viral replication and HIV-1 strains resistant to multiple antiretroviral agents. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4hy5_ligand_3_34.mol2 | 4hy5 | 0.894737 | -6.34 | C(C1CCC(F)(F)CC1)NC(=O)CC | 14 |
| 4hy0_ligand_3_34.mol2 | 4hy0 | 0.894737 | -6.23 | C(=O)(NCC1CCC(F)(F)CC1)CC | 14 |
| 3hd3_ligand_5_407.mol2 | 3hd3 | 0.864865 | -7.05 | C[C@H](CC)NC(=O)C1CCCCC1 | 13 |
| 3hd3_ligand_5_338.mol2 | 3hd3 | 0.864865 | -7.00 | CC[C@H](C)NC(=O)C1CCCCC1 | 13 |
| 3hd3_ligand_4_296.mol2 | 3hd3 | 0.864865 | -6.84 | CCCNC(=O)C1CCCCC1 | 12 |
| 3hd3_ligand_4_212.mol2 | 3hd3 | 0.864865 | -6.73 | CCCNC(=O)C1CCCCC1 | 12 |
| 4bak_ligand_2_26.mol2 | 4bak | 0.842105 | -7.17 | C(=O)(N1CCC1)CC1CCCCC1 | 13 |
| 4dmy_ligand_2_0.mol2 | 4dmy | 0.842105 | -7.03 | C1(CCCCC1)C(=O)NC1CC1 | 12 |
| 4tw7_ligand_2_121.mol2 | 4tw7 | 0.820513 | -8.36 | C1CCCCN1C(=O)CC1CCCCC1 | 15 |
129 ,
13

