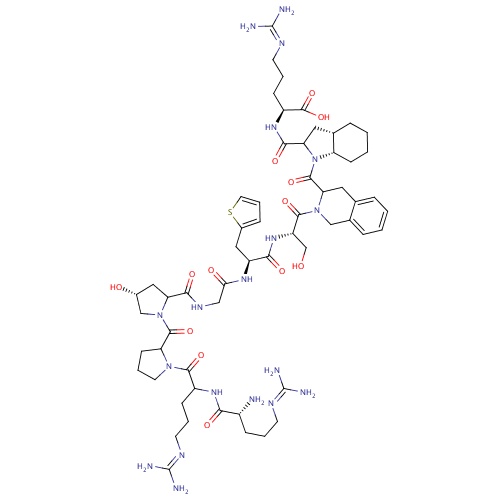
IUPAC name
(2S)-2-{[(3aS,7aS)-1-{2-[(2S)-2-[(2S)-2-(2-{[(4R)-1-(1-{2-[(2R)-2-amino-5-[(diaminomethylidene)amino]pentanamido]-5-[(diaminomethylidene)amino]pentanoyl}pyrrolidine-2-carbonyl)-4-hydroxypyrrolidin-2-yl]formamido}acetamido)-3-(thiophen-2-yl)propanamido]-3-hydroxypropanoyl]-1,2,3,4-tetrahydroisoquinoline-3-carbonyl}-octahydro-1H-indol-2-yl]formamido}-5-[(diaminomethylidene)amino]pentanoic acid
SMILES
[H][C@]12CC(N(C(=O)C3CC4=CC=CC=C4CN3C(=O)[C@H](CO)NC(=O)[C@H](CC3=CC=CS3)NC(=O)CNC(=O)C3C[C@@H](O)CN3C(=O)C3CCCN3C(=O)C(CCCN=C(N)N)NC(=O)[C@H](N)CCCN=C(N)N)[C@@]1([H])CCCC2)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O
Compound class
Anti-Inflammatory Agents, Non-Steroidal; Adrenergic beta-Antagonists; Blood and Blood Forming Organs; Drugs Used in Hereditary Angioedema; Bradykinin B2 Receptor Antagonists;
Therapeutic area
Approved for use in acute attacks of hereditary angioedema (HAE). Investigated for use/treatment in angioedema, liver disease, and burns and burn infections.
Common name
Icatibant
IUPAC name
(2S)-2-{[(3aS,7aS)-1-{2-[(2S)-2-[(2S)-2-(2-{[(4R)-1-(1-{2-[(2R)-2-amino-5-[(diaminomethylidene)amino]pentanamido]-5-[(diaminomethylidene)amino]pentanoyl}pyrrolidine-2-carbonyl)-4-hydroxypyrrolidin-2-yl]formamido}acetamido)-3-(thiophen-2-yl)propanamido]-3-hydroxypropanoyl]-1,2,3,4-tetrahydroisoquinoline-3-carbonyl}-octahydro-1H-indol-2-yl]formamido}-5-[(diaminomethylidene)amino]pentanoic acid
SMILES
[H][C@]12CC(N(C(=O)C3CC4=CC=CC=C4CN3C(=O)[C@H](CO)NC(=O)[C@H](CC3=CC=CS3)NC(=O)CNC(=O)C3C[C@@H](O)CN3C(=O)C3CCCN3C(=O)C(CCCN=C(N)N)NC(=O)[C@H](N)CCCN=C(N)N)[C@@]1([H])CCCC2)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O
INCHI
InChI=1S/C59H89N19O13S/c60-37(14-5-19-67-57(61)62)48(82)72-38(15-6-20-68-58(63)64)52(86)75-22-8-18-43(75)54(88)77-30-35(80)26-44(77)50(84)70-28-47(81)71-40(27-36-13-9-23-92-36)49(83)74-41(31-79)53(87)76-29-34-12-2-1-10-32(34)24-46(76)55(89)78-42-17-4-3-11-33(42)25-45(78)51(85)73-39(56(90)91)16-7-21-69-59(65)66/h1-2,9-10,12-13,23,33,35,37-46,79-80H,3-8,11,14-22,24-31,60H2,(H,70,84)(H,71,81)(H,72,82)(H,73,85)(H,74,83)(H,90,91)(H4,61,62,67)(H4,63,64,68)(H4,65,66,69)/t33-,35+,37+,38?,39-,40-,41-,42-,43?,44?,45?,46?/m0/s1
FORMULA
C59H89N19O13S

Common name
Icatibant
IUPAC name
(2S)-2-{[(3aS,7aS)-1-{2-[(2S)-2-[(2S)-2-(2-{[(4R)-1-(1-{2-[(2R)-2-amino-5-[(diaminomethylidene)amino]pentanamido]-5-[(diaminomethylidene)amino]pentanoyl}pyrrolidine-2-carbonyl)-4-hydroxypyrrolidin-2-yl]formamido}acetamido)-3-(thiophen-2-yl)propanamido]-3-hydroxypropanoyl]-1,2,3,4-tetrahydroisoquinoline-3-carbonyl}-octahydro-1H-indol-2-yl]formamido}-5-[(diaminomethylidene)amino]pentanoic acid
Molecular weight
1304.522
clogP
-1.081
clogS
-3.483
HBond Acceptor
16
HBond Donor
22
Total Polar Surface Area
551.96
Number of Rings
7
Rotatable Bond
34
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00003 | formamide |
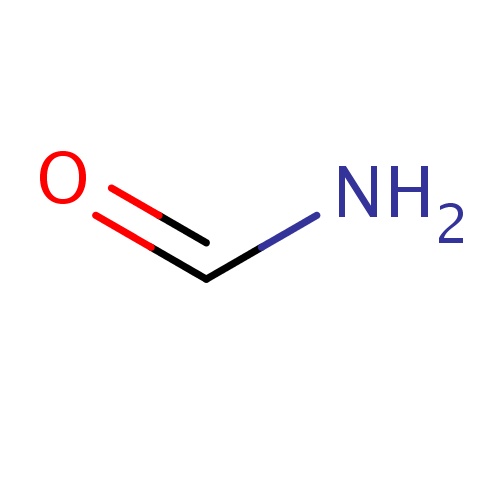
|
C(=O)N | 0.1240 |
| FDBF00004 | acetic acid |
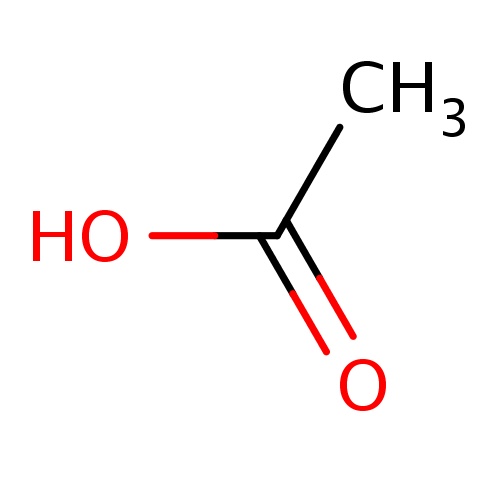
|
CC(=O)O | 0.0687 |
| FDBF00009 | pyrrole-1-carbaldehyde |
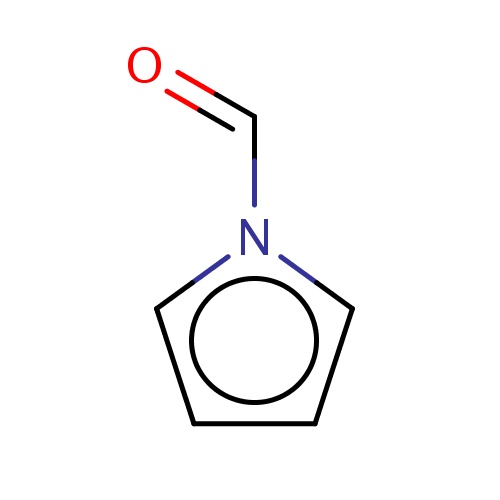
|
O=Cn1cccc1 | 0.0021 |
| FDBF00660 | thiophene |
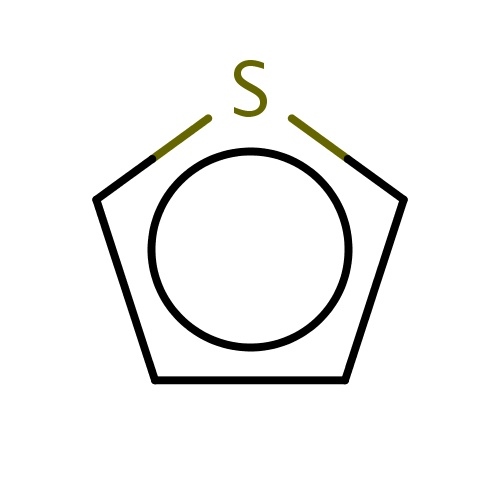
|
s1cccc1 | 0.0076 |
| FDBF03355 | (3aS,7aS)-2,3,3a,4,5,6,7,7a-octahydroindole-1-carbaldehyde |
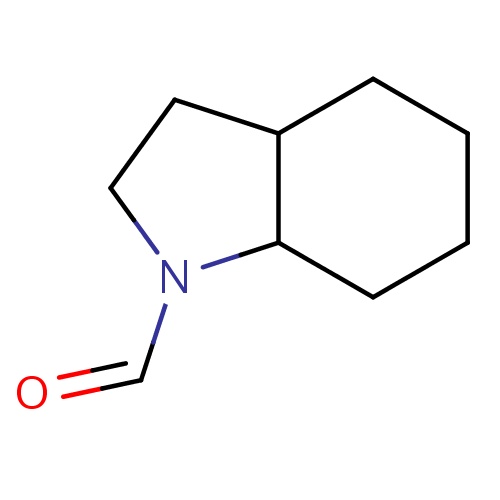
|
O=CN1C2C(CC1)CCCC2 | 0.0003 |
| FDBF03356 | 3,4-dihydro-1H-isoquinoline-2-carbaldehyde |
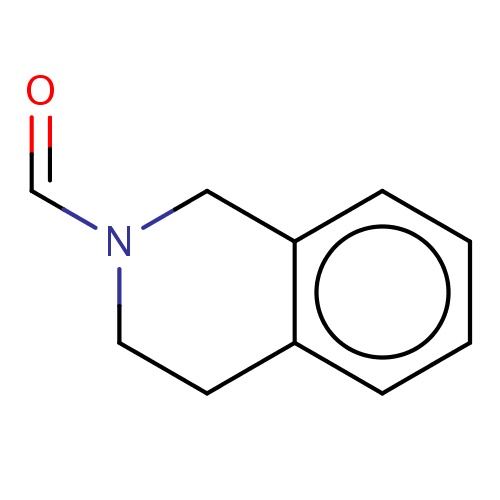
|
O=CN1CCc2c(cccc2)C1 | 0.0003 |
| FDBF03357 | (3R)-3-hydroxypyrrolidine-1-carbaldehyde |
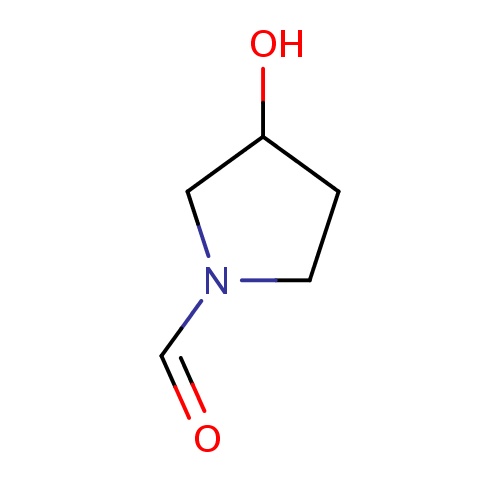
|
OC1CCN(C1)C=O | 0.0003 |

