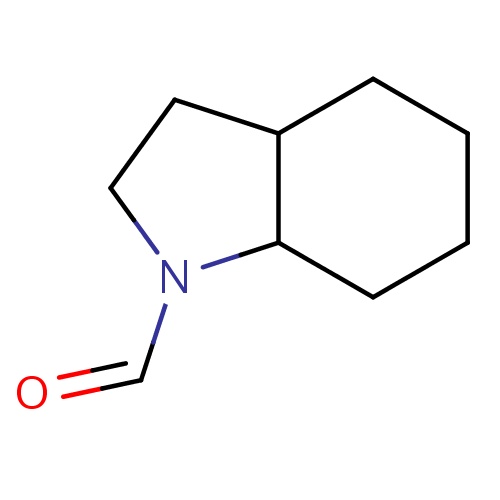
Common name
(3aS,7aS)-2,3,3a,4,5,6,7,7a-octahydroindole-1-carbaldehyde
IUPAC name
(3aS,7aS)-2,3,3a,4,5,6,7,7a-octahydroindole-1-carbaldehyde
SMILES
O=CN1C2C(CC1)CCCC2
Common name
(3aS,7aS)-2,3,3a,4,5,6,7,7a-octahydroindole-1-carbaldehyde
IUPAC name
(3aS,7aS)-2,3,3a,4,5,6,7,7a-octahydroindole-1-carbaldehyde
SMILES
O=CN1C2C(CC1)CCCC2
INCHI
InChI=1S/C9H15NO/c11-7-10-6-5-8-3-1-2-4-9(8)10/h7-9H,1-6H2/t8-,9-/m0/s1
FORMULA
C9H15NO

Common name
(3aS,7aS)-2,3,3a,4,5,6,7,7a-octahydroindole-1-carbaldehyde
IUPAC name
(3aS,7aS)-2,3,3a,4,5,6,7,7a-octahydroindole-1-carbaldehyde
Molecular weight
153.221
clogP
1.439
clogS
-0.905
Frequency
0.0003
HBond Acceptor
1
HBond Donor
0
Total PolarSurface Area
20.31
Number of Rings
2
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
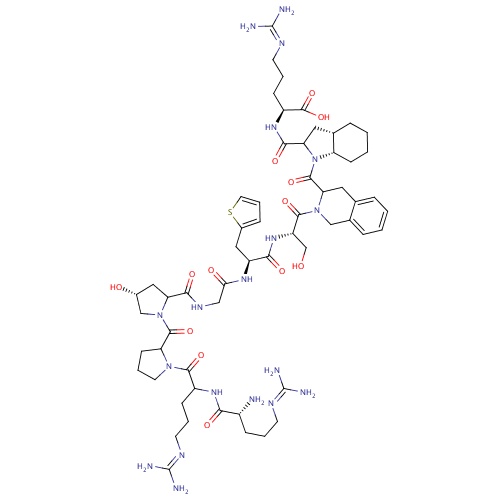
| FDBD01381 | Icatibant |
|
Anti-Inflammatory Agents, Non-Steroidal; Adrenergic beta-Antagonists; Blood and Blood Forming Organs; Drugs Used in Hereditary Angioedema; Bradykinin B2 Receptor Antagonists; | Approved for use in acute attacks of hereditary angioedema (HAE). Investigated for use/treatment in angioedema, liver disease, and burns and burn infections. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3bv9_ligand_frag_4.mol2 | 3bv9 | 1 | -6.63 | C(=O)N1CC[C@H]2[C@@H]1CCCC2 | 11 |
| 1aq7_ligand_frag_7.mol2 | 1aq7 | 1 | -6.11 | C(=O)N1CC[C@H]2[C@@H]1CCCC2 | 11 |
| 2p59_ligand_frag_0.mol2 | 2p59 | 1 | -5.60 | [C@H]12[C@H](CCCC1)N(CC2)C=O | 11 |
| 1aq7_ligand_1_4.mol2 | 1aq7 | 0.966667 | -6.22 | C(=O)(N1CC[C@H]2[C@@H]1CCCC2)C | 12 |
| 2p59_ligand_1_4.mol2 | 2p59 | 0.966667 | -5.78 | CC(=O)N1[C@@H]2[C@@H](CCCC2)CC1 | 12 |
| 4twy_ligand_1_5.mol2 | 4twy | 0.933333 | -7.16 | C[C@@H]1C[C@H]2CCCC[C@@H]2CN1C=O | 13 |
| 4tww_ligand_1_4.mol2 | 4tww | 0.933333 | -7.11 | C[C@@H]1C[C@@H]2[C@@H](CN1C=O)CCCC2 | 13 |
| 4wy3_ligand_1_3.mol2 | 4wy3 | 0.933333 | -7.04 | [C@@H]1(C[C@@H]2CCCC[C@H]2CN1C=O)C | 13 |
| 4twy_ligand_frag_6.mol2 | 4twy | 0.933333 | -6.86 | C1C[C@H]2CCCC[C@@H]2CN1C=O | 12 |
| 4tww_ligand_frag_1.mol2 | 4tww | 0.933333 | -6.81 | C1[C@@H]2[C@@H](CCN1C=O)CCCC2 | 12 |
107 ,
11

