
IUPAC name
2-chloro-N-[4-chloro-3-(pyridin-2-yl)phenyl]-4-methanesulfonylbenzamide
SMILES
CS(=O)(=O)C1=CC(Cl)=C(C=C1)C(=O)NC1=CC=C(Cl)C(=C1)C1=CC=CC=N1
Compound class
Antineoplastic Agents; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors;
Therapeutic area
Vismodegib is used for treating locally advanced or metastatic basal cell carcinoma in patients whose carcinoma has recurred after surgery, and in patients who are not candidates for surgery or radiation.
Common name
Vismodegib
IUPAC name
2-chloro-N-[4-chloro-3-(pyridin-2-yl)phenyl]-4-methanesulfonylbenzamide
SMILES
CS(=O)(=O)C1=CC(Cl)=C(C=C1)C(=O)NC1=CC=C(Cl)C(=C1)C1=CC=CC=N1
INCHI
InChI=1S/C19H14Cl2N2O3S/c1-27(25,26)13-6-7-14(17(21)11-13)19(24)23-12-5-8-16(20)15(10-12)18-4-2-3-9-22-18/h2-11H,1H3,(H,23,24)
FORMULA
C19H14Cl2N2O3S

Common name
Vismodegib
IUPAC name
2-chloro-N-[4-chloro-3-(pyridin-2-yl)phenyl]-4-methanesulfonylbenzamide
Molecular weight
421.297
clogP
4.238
clogS
-6.993
HBond Acceptor
4
HBond Donor
1
Total Polar Surface Area
76.13
Number of Rings
3
Rotatable Bond
4
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00003 | formamide |
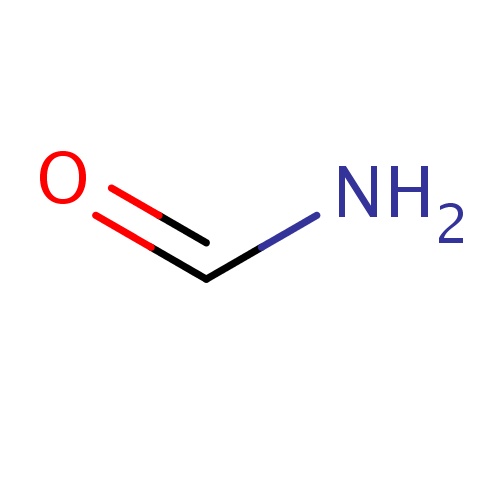
|
C(=O)N | 0.1240 |
| FDBF00014 | pyridine |
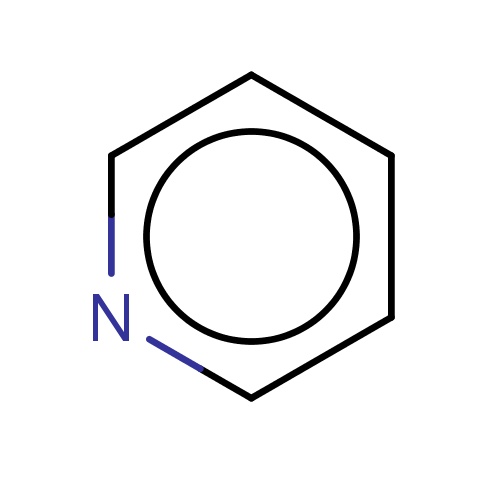
|
c1cccnc1 | 0.0333 |
| FDBF00016 | chlorobenzene |
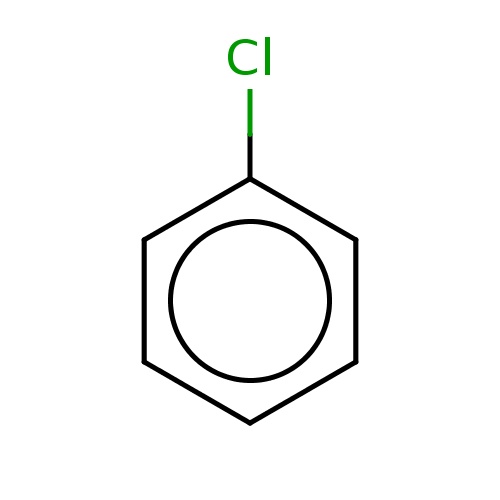
|
c1ccc(cc1)Cl | 0.0718 |
| FDBF00317 | (dihydroxy-λ3-sulfanyl)methane |
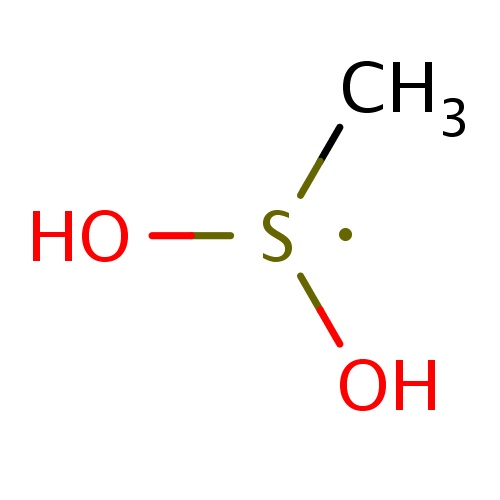
|
C[S](O)O | 0.0141 |
| FDBF03730 | 1-chloro-3-methylsulfonyl-benzene |
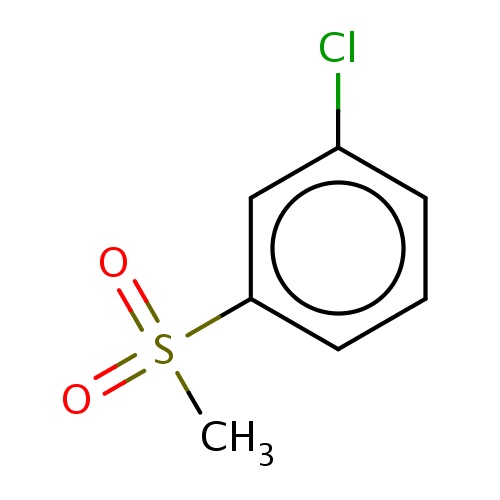
|
Clc1cccc(c1)S(=O)(=O)C | 0.0024 |
| FDBF03732 | 2-chlorobenzamide |
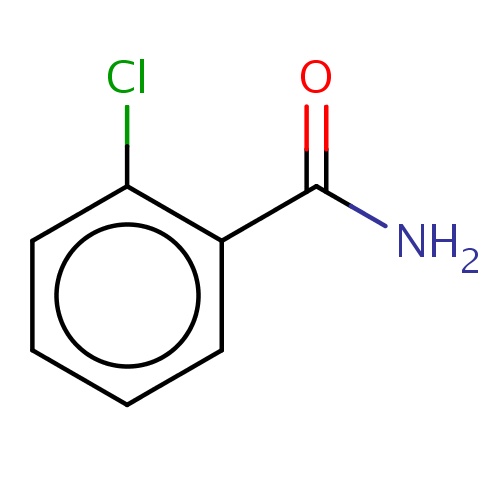
|
Clc1c(cccc1)C(=O)N | 0.0007 |
| FDBF03733 | N-[4-chloro-3-(2-pyridyl)phenyl]formamide |
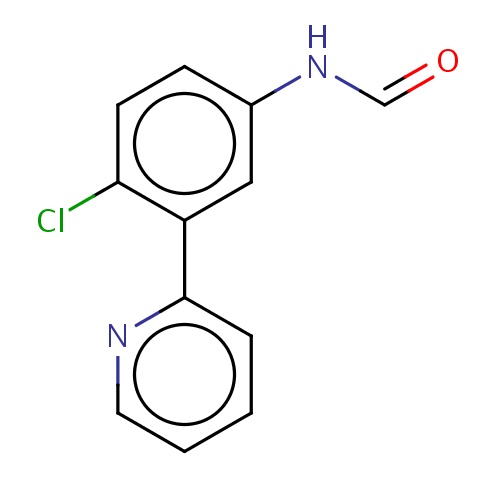
|
n1c(cccc1)c2c(ccc(c2)NC=O)Cl | 0.0003 |

