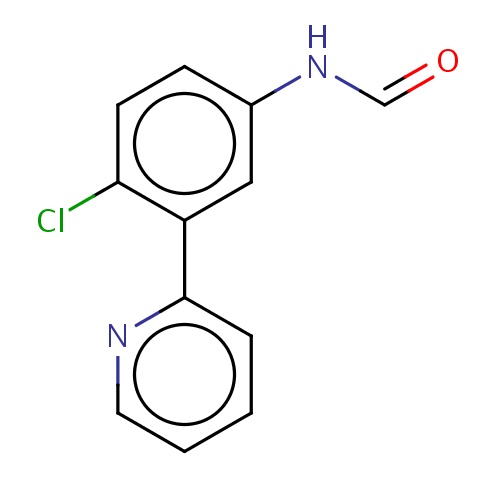
Common name
N-[4-chloro-3-(2-pyridyl)phenyl]formamide
IUPAC name
N-[4-chloro-3-(2-pyridyl)phenyl]formamide
SMILES
n1c(cccc1)c2c(ccc(c2)NC=O)Cl
Common name
N-[4-chloro-3-(2-pyridyl)phenyl]formamide
IUPAC name
N-[4-chloro-3-(2-pyridyl)phenyl]formamide
SMILES
n1c(cccc1)c2c(ccc(c2)NC=O)Cl
INCHI
InChI=1S/C12H9ClN2O/c13-11-5-4-9(15-8-16)7-10(11)12-3-1-2-6-14-12/h1-8H,(H,15,16)
FORMULA
C12H9ClN2O

Common name
N-[4-chloro-3-(2-pyridyl)phenyl]formamide
IUPAC name
N-[4-chloro-3-(2-pyridyl)phenyl]formamide
Molecular weight
232.666
clogP
2.974
clogS
-4.106
Frequency
0.0003
HBond Acceptor
2
HBond Donor
1
Total PolarSurface Area
41.99
Number of Rings
2
Rotatable Bond
2
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01538 | Vismodegib |

|
Antineoplastic Agents; Antineoplastic and Immunomodulating Agents; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; | Vismodegib is used for treating locally advanced or metastatic basal cell carcinoma in patients whose carcinoma has recurred after surgery, and in patients who are not candidates for surgery or radiation. |
1 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 4f4p_ligand_2_0.mol2 | 4f4p | 0.604167 | -7.63 | c1(cc(ccc1)c1ncccc1)N1CC[NH2+]CC1 | 18 |
| 1w83_ligand_2_2.mol2 | 1w83 | 0.530864 | -7.47 | Cc1c(Cl)ccc(c1)NC=O | 11 |
| 3mw1_ligand_1_2.mol2 | 3mw1 | 0.521368 | -8.03 | c1c2c(ncc1)c([n+](cc2)O)c1c(cccc1Cl)Cl | 19 |
| 4ql1_ligand_2_10.mol2 | 4ql1 | 0.518519 | -7.73 | C(=O)Nc1cc(ccc1)c1ccccc1 | 15 |
| 4f4p_ligand_2_5.mol2 | 4f4p | 0.505618 | -7.17 | Nc1nc(c2ccccc2)ccc1 | 13 |
| 4uvv_ligand.mol2 | 4uvv | 0.5 | -10.00 | c1c2c(C)cccc2c(=O)[nH]c1c1ccc(Cl)cc1 | 20 |
| 2aqu_ligand_4_1774.mol2 | 2aqu | 0.5 | -6.91 | c1(ccc(cc1)c1ccccn1)/C=[NH]/C | 15 |
| 2wkz_ligand_3_52.mol2 | 2wkz | 0.488636 | -6.99 | [NH3+]Cc1ccc(c2ncccc2)cc1 | 14 |
| 2o4k_ligand_3_549.mol2 | 2o4k | 0.488636 | -6.94 | [NH3+]Cc1ccc(cc1)c1ccccn1 | 14 |
| 2cem_ligand_3_203.mol2 | 2cem | 0.488636 | -6.92 | C([NH3+])c1ccc(c2ccccn2)cc1 | 14 |
102 ,
11

