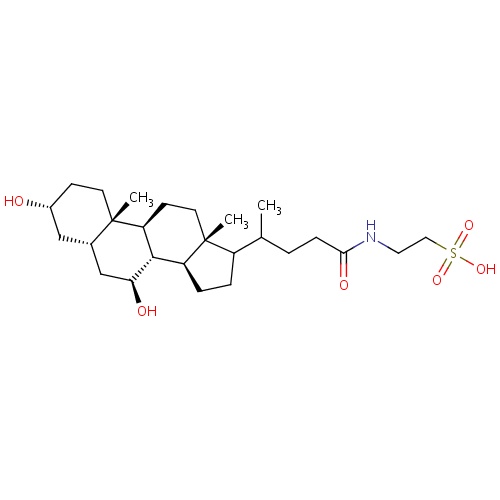
IUPAC name
2-{4-[(1S,2S,5R,7S,9S,10R,11S,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,
SMILES
[H][C@@]12CCC(C(C)CCC(=O)NCCS(O)(=O)=O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@@H](O)C[C@]2([H])C[C@H](O)CC[C@]12C
Compound class
Antiviral Agents; Cholagogues and Choleretics; Cholesterol Absorption Inhibitors;
Therapeutic area
Used in the treatment of cholesterol gallstones. Tauroursodeoxycholic acid is also being investigated for use in several conditions such as Primary Biliary Cirrhosis (PBC), insulin resistance, amyloidosis, Cystic Fibrosis, Cholestasis, and Amyotrophic Lateral Sclerosis.
Common name
Tauroursodeoxycholic acid
IUPAC name
2-{4-[(1S,2S,5R,7S,9S,10R,11S,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,
SMILES
[H][C@@]12CCC(C(C)CCC(=O)NCCS(O)(=O)=O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@@H](O)C[C@]2([H])C[C@H](O)CC[C@]12C
INCHI
InChI=1S/C26H45NO6S/c1-16(4-7-23(30)27-12-13-34(31,32)33)19-5-6-20-24-21(9-11-26(19,20)3)25(2)10-8-18(28)14-17(25)15-22(24)29/h16-22,24,28-29H,4-15H2,1-3H3,(H,27,30)(H,31,32,33)/t16?,17-,18+,19?,20-,21-,22-,24-,25-,26+/m0/s1
FORMULA
C26H45NO6S

Common name
Tauroursodeoxycholic acid
IUPAC name
2-{4-[(1S,2S,5R,7S,9S,10R,11S,15R)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0²,
Molecular weight
499.704
clogP
2.321
clogS
-3.811
HBond Acceptor
6
HBond Donor
4
Total Polar Surface Area
123.93
Number of Rings
4
Rotatable Bond
7
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
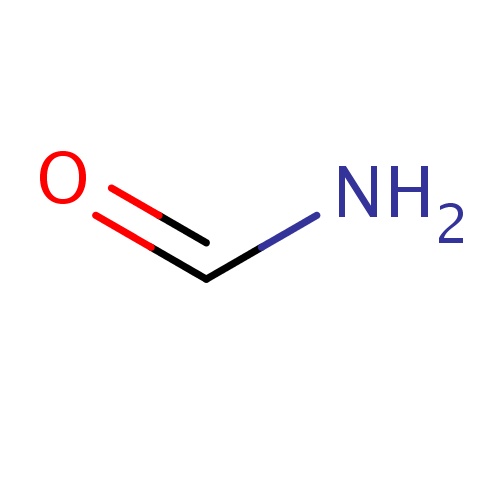
| FDBF00003 | formamide |
|
C(=O)N | 0.1240 |
| FDBF00007 | propane |

|
C(C)C | 0.2412 |
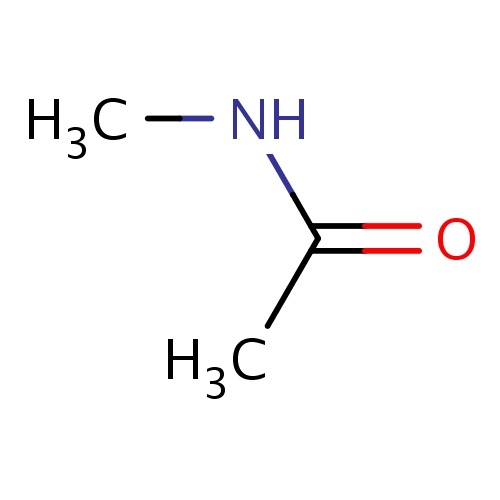
| FDBF00027 | N-methylacetamide |
|
N(C(=O)C)C | 0.0175 |
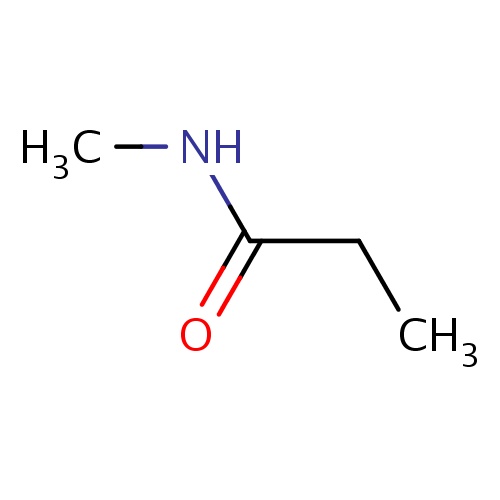
| FDBF00031 | N-methylpropanamide |
|
N(C(=O)CC)C | 0.0069 |
| FDBF00067 | butane |
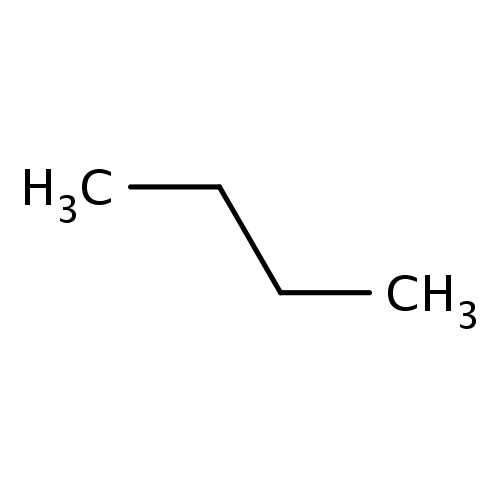
|
CCCC | 0.0680 |
| FDBF00185 | pentanamide |
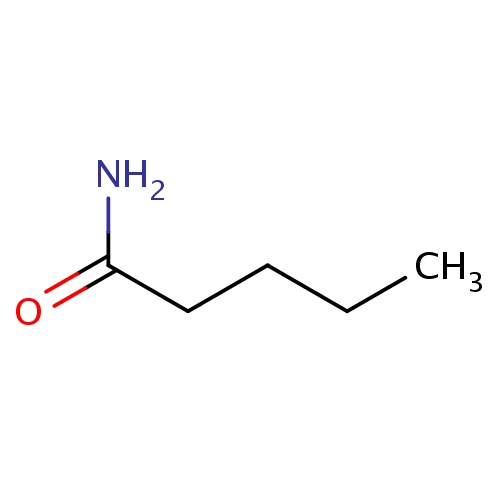
|
C(CC(=O)N)CC | 0.0024 |
| FDBF00187 | N-methylpentanamide |
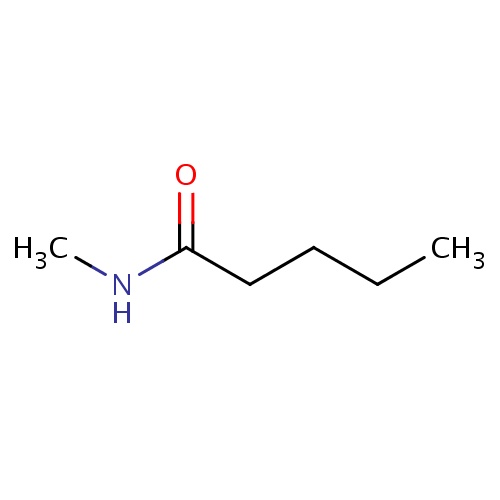
|
C(CC(=O)NC)CC | 0.0017 |
| FDBF00228 | N-ethylacetamide |
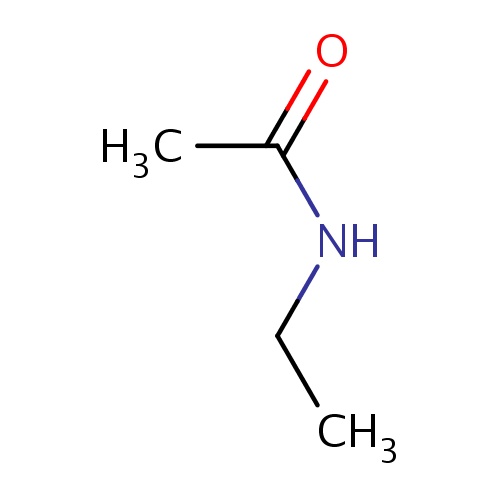
|
C(=O)(NCC)C | 0.0089 |
| FDBF00234 | N-ethylpropanamide |

|
C(=O)(NCC)CC | 0.0038 |
| FDBF01505 | methanesulfonic acid |
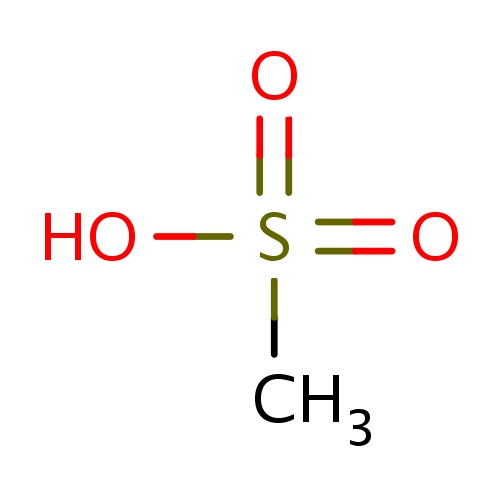
|
CS(=O)(=O)O | 0.0038 |

