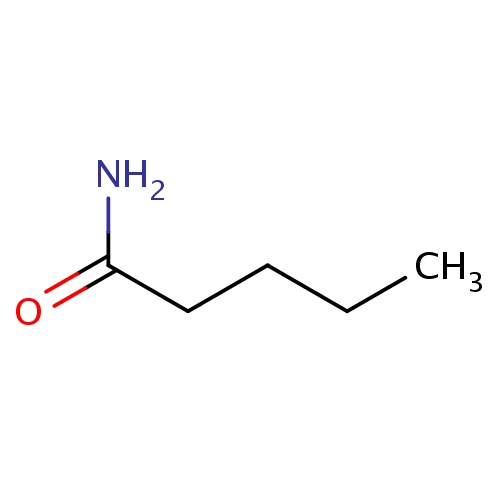
Common name
pentanamide
IUPAC name
pentanamide
SMILES
C(CC(=O)N)CC
Common name
pentanamide
IUPAC name
pentanamide
SMILES
C(CC(=O)N)CC
INCHI
InChI=1S/C5H11NO/c1-2-3-4-5(6)7/h2-4H2,1H3,(H2,6,7)
FORMULA
C5H11NO

Common name
pentanamide
IUPAC name
pentanamide
Molecular weight
101.147
clogP
0.284
clogS
-0.994
Frequency
0.0024
HBond Acceptor
1
HBond Donor
2
Total PolarSurface Area
43.09
Number of Rings
0
Rotatable Bond
3
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD00066 | Valsartan |
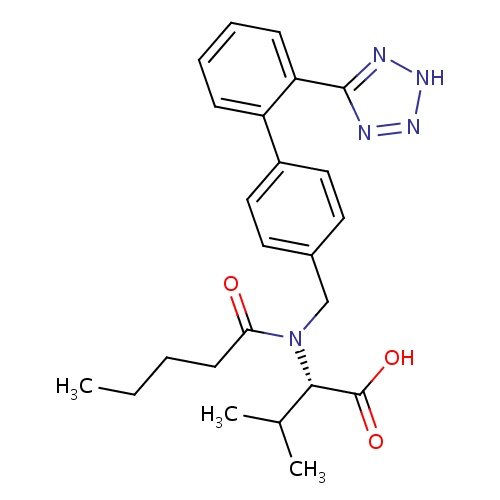
|
Antihypertensive Agents; Angiotensin II Type 1 Receptor Blockers; Angiotensin Receptor Antagonists; Lipid Modifying Agents; Cardiovascular System; Angiotensin II Antagonists, Plain; Agents Acting on the Renin-Angiotensin System; Angiotensin II Antagonists and Diuretics; Angiotensin II Antagonists and Calcium Channel Blockers; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Angiotensin II Receptor Antagonists; | May be used as a first line agent to treat uncomplicated hypertension, isolated systolic hypertension and left ventricular hypertrophy. May be used as a first line agent to delay progression of diabetic nephropathy. Losartan may be also used as a second line agent in the treatment of congestive heart failure, systolic dysfunction, myocardial infarction and coronary artery disease in those intolerant of ACE inhibitors. |
| FDBD01287 | Vorinostat |
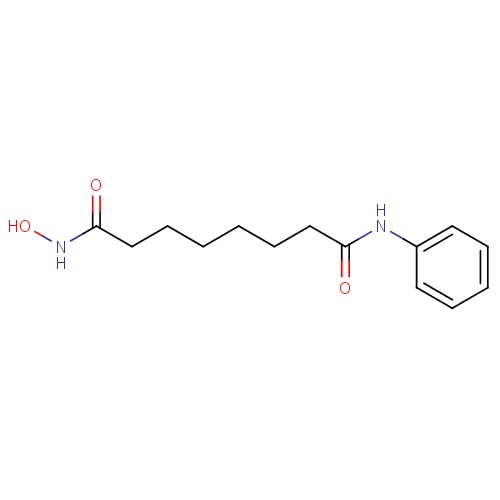
|
Antineoplastic Agents; Histone Deacetylase Inhibitors; Antineoplastic and Immunomodulating Agents; | For the treatment of cutaneous manifestations in patients with cutaneous T-cell lymphoma who have progressive, persistent or recurrent disease on or following two systemic therapies. |
| FDBD01317 | Iodipamide |
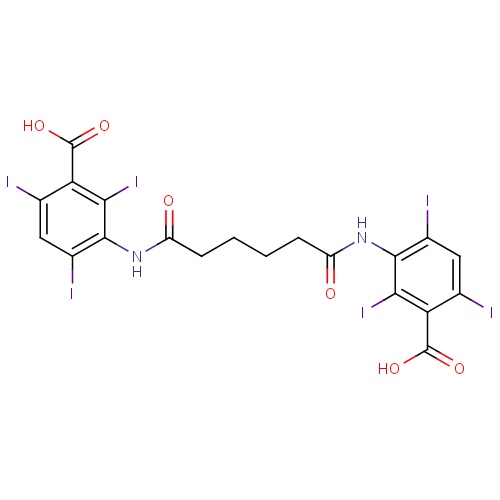
|
Contrast Media; X-Ray Contrast Media, Iodinated; Watersoluble, Hepatotropic X-Ray Contrast Media; | Iodipamide is used as a contrast agent for cholecystography and intravenous cholangiography. |
| FDBD01475 | Capsaicin |
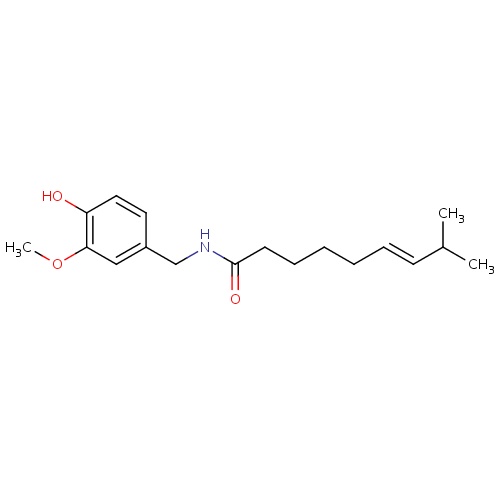
|
Anesthetics, Local; Antipruritics; Anesthetics; Sensory System Agents; Musculo-Skeletal System; Nervous System; Topical Products for Joint and Muscular Pain; Capsaicin and Similar Agents; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; CYP2E1 Inhibitors; CYP2E1 Inducers; CYP2E1 Inducers (strong); CYP3A4 Inhibitors; | The capsaicin 8% patch is indicated in the treatment of neuropathic pain associated with post-herpetic neuralgia. There are multiple topical capsaicin formulations available, including creams and solutions, indicated for temporary analgesia in muscle and join pain as well as neuropathic pain. |
| FDBD01539 | Tauroursodeoxycholic acid |
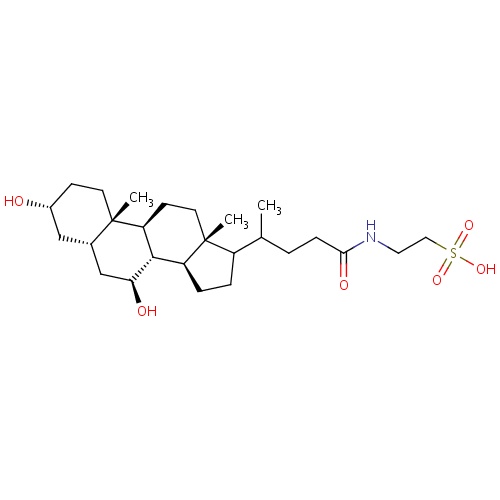
|
Antiviral Agents; Cholagogues and Choleretics; Cholesterol Absorption Inhibitors; | Used in the treatment of cholesterol gallstones. Tauroursodeoxycholic acid is also being investigated for use in several conditions such as Primary Biliary Cirrhosis (PBC), insulin resistance, amyloidosis, Cystic Fibrosis, Cholestasis, and Amyotrophic Lateral Sclerosis. |
| FDBD01640 | Eliglustat |
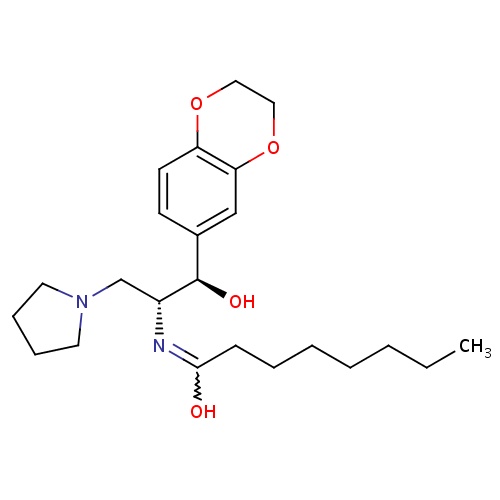
|
Enzyme Inhibitors; Alimentary Tract and Metabolism; Various Alimentary Tract and Metabolism Products; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | Eliglustat is indicated for the long-term treatment of type 1 Gaucher disease in patients who are CYP2D6 extensive metabolizers (EMs), intermediate metabolizers (IMs), or poor metabolizers (PMs) in treatment-naive and treatment-experienced adult patients. |
| FDBD01692 | Zucapsaicin |
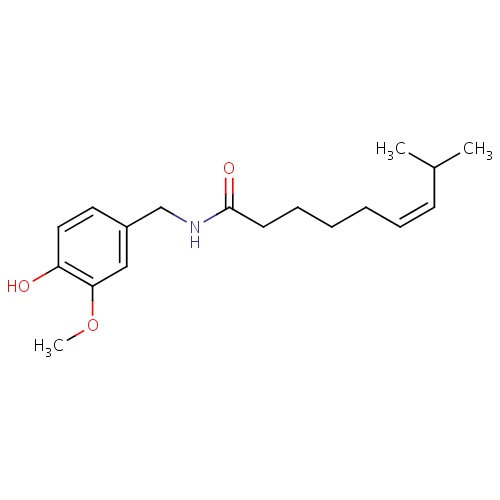
|
Musculo-Skeletal System; Topical Products for Joint and Muscular Pain; Capsaicin and Similar Agents; |
7 ,
1
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3g0i_ligand_4_0.mol2 | 3g0i | 1 | -6.85 | C[C@H](CCC)C(=O)N | 8 |
| 3g0i_ligand_4_4.mol2 | 3g0i | 1 | -6.84 | C([C@H](C)C(=O)N)CC | 8 |
| 1mnc_ligand_4_56.mol2 | 1mnc | 1 | -6.78 | C([C@H](C)C(=O)N)C(C)C | 9 |
| 1mmr_ligand_4_11.mol2 | 1mmr | 1 | -6.77 | C(=O)(N)[C@@H](C)CC(C)C | 9 |
| 1rm8_ligand_4_334.mol2 | 1rm8 | 1 | -6.74 | C(C)(C)C[C@@H](C(=O)N)C | 9 |
| 1y3g_ligand_4_680.mol2 | 1y3g | 1 | -6.74 | C[C@H](C(=O)N)CC(C)C | 9 |
| 1mmq_ligand_4_11.mol2 | 1mmq | 1 | -6.73 | C(C(C)C)[C@H](C)C(=O)N | 9 |
| 1biw_ligand_4_289.mol2 | 1biw | 1 | -6.69 | NC(=O)[C@@H](C)CC(C)C | 9 |
| 2w12_ligand_4_175.mol2 | 2w12 | 1 | -6.66 | [C@@H](CC(C)C)(C)C(=O)N | 9 |
371 ,
38

