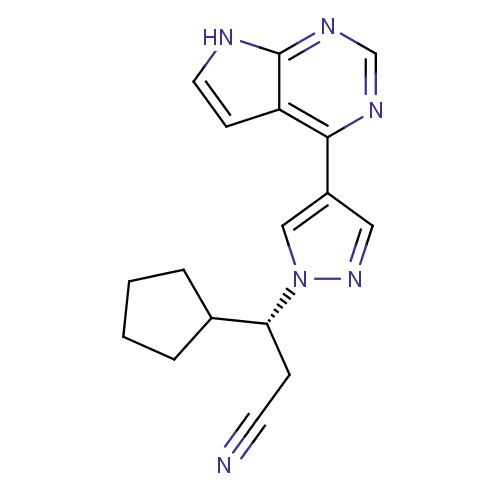
IUPAC name
(3R)-3-cyclopentyl-3-(4-{7H-pyrrolo[2,3-d]pyrimidin-4-yl}-1H-pyrazol-1-yl)propanenitrile
SMILES
N#CC[C@H](C1CCCC1)N1C=C(C=N1)C1=C2C=CNC2=NC=N1
Compound class
Antineoplastic Agents; Immunosuppressive Agents; Protein Kinase Inhibitors; Antineoplastic and Immunomodulating Agents; CYP3A4 Inhibitors;
Therapeutic area
Treatment of intermediate or high-risk myelofibrosis, including primary myelofibrosis, post-polycythemia vera (post-PV) myelofibrosis and post-essential thrombocythemia (post-ET) myelofibrosis. [Lexicomp] Myeolofibrosis is the proliferation of abnormal bone marrow stem cells which cause fibrosis (the excessive formation of connective tissue).
Common name
Ruxolitinib
IUPAC name
(3R)-3-cyclopentyl-3-(4-{7H-pyrrolo[2,3-d]pyrimidin-4-yl}-1H-pyrazol-1-yl)propanenitrile
SMILES
N#CC[C@H](C1CCCC1)N1C=C(C=N1)C1=C2C=CNC2=NC=N1
INCHI
InChI=1S/C17H18N6/c18-7-5-15(12-3-1-2-4-12)23-10-13(9-22-23)16-14-6-8-19-17(14)21-11-20-16/h6,8-12,15H,1-5H2,(H,19,20,21)/t15-/m1/s1
FORMULA
C17H18N6

Common name
Ruxolitinib
IUPAC name
(3R)-3-cyclopentyl-3-(4-{7H-pyrrolo[2,3-d]pyrimidin-4-yl}-1H-pyrazol-1-yl)propanenitrile
Molecular weight
306.365
clogP
2.743
clogS
-3.995
HBond Acceptor
4
HBond Donor
1
Total Polar Surface Area
83.18
Number of Rings
4
Rotatable Bond
5
| Drug ID | Common name | Structure CAS | SMILE | Frequency |
|---|---|---|---|---|
| FDBF00567 | 1H-pyrazole |

|
[nH]1nccc1 | 0.0021 |
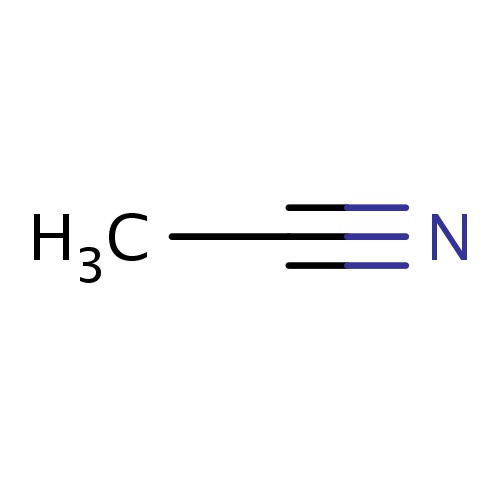
| FDBF00569 | acetonitrile |
|
CC#N | 0.0161 |
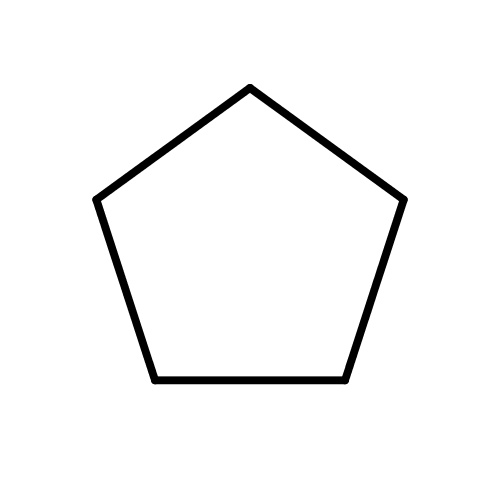
| FDBF01245 | cyclopentane |
|
C1CCCC1 | 0.0034 |
| FDBF01402 | methylcyclopentane |

|
CC1CCCC1 | 0.0007 |
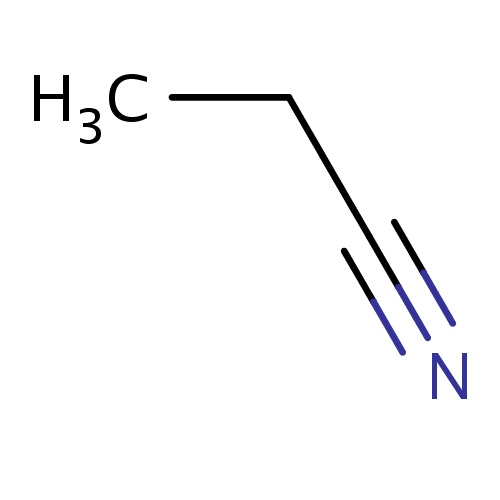
| FDBF01513 | propanenitrile |
|
C(C#N)C | 0.0048 |
| FDBF03759 | 1-methylpyrazole |

|
n1(nccc1)C | 0.0003 |
| FDBF03760 | 1-(cyclopentylmethyl)pyrazole |

|
n1(nccc1)CC2CCCC2 | 0.0003 |
| FDBF03761 | 3-pyrazol-1-ylpropanenitrile |

|
C(C#N)Cn1nccc1 | 0.0003 |
| FDBF03762 | 3-cyclopentylpropanenitrile |

|
C(C#N)CC1CCCC1 | 0.0003 |
| FDBF03763 | 7H-pyrrolo[2,3-d]pyrimidine |

|
n1cc2c([nH]cc2)nc1 | 0.0007 |

