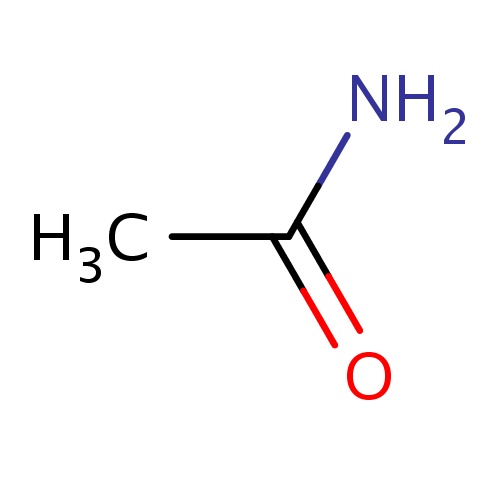
Common name
acetamide
IUPAC name
acetamide
SMILES
NC(=O)C
Common name
acetamide
IUPAC name
acetamide
SMILES
NC(=O)C
INCHI
InChI=1S/C2H5NO/c1-2(3)4/h1H3,(H2,3,4)
FORMULA
C2H5NO

Common name
acetamide
IUPAC name
acetamide
Molecular weight
59.067
clogP
-0.723
clogS
0.321
Frequency
0.0460
HBond Acceptor
1
HBond Donor
2
Total PolarSurface Area
43.09
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
| FDBD01609 | Etidocaine |

|
Anesthetics, Local; Anesthetics; Nervous System; Amides; | |
| FDBD01613 | Enviomycin |
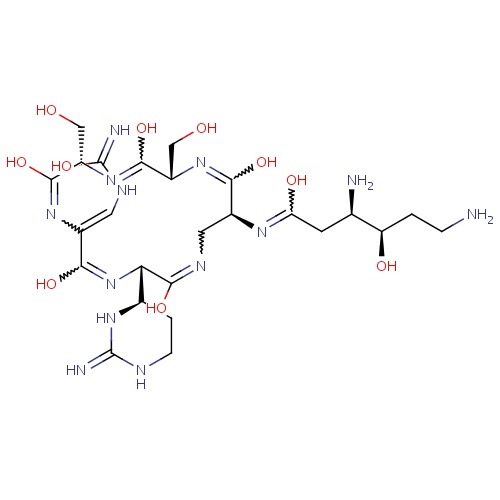
|
Anti-Bacterial Agents; Antibiotics, Antitubercular; | |
| FDBD01622 | Cephaloridine |
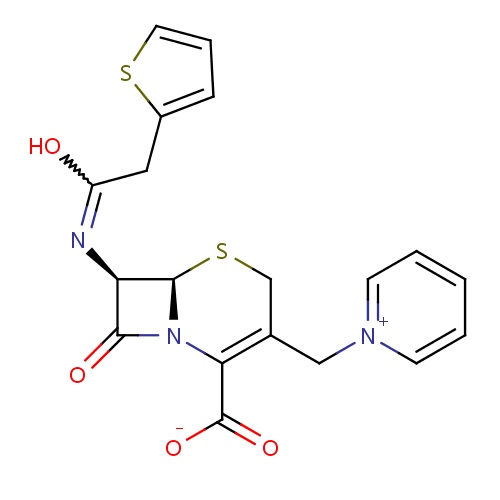
|
Anti-Bacterial Agents; | |
| FDBD01633 | Aliskiren |
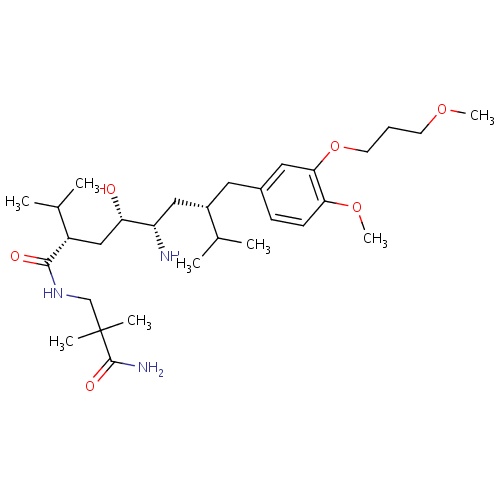
|
Cardiovascular System; Agents Acting on the Renin-Angiotensin System; Renin-Inhibitors; CYP3A4 Inhibitors; | For the treatment of hypertension, to lower blood pressure. |
| FDBD01640 | Eliglustat |
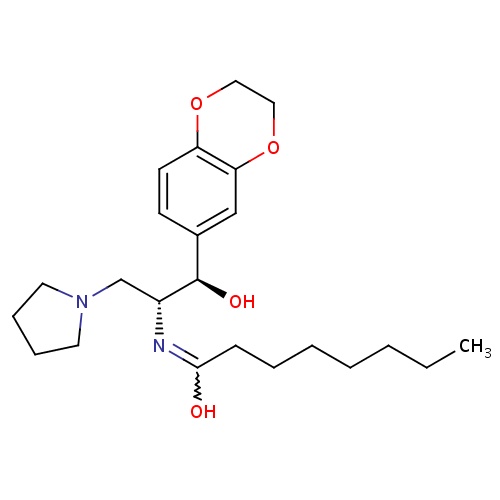
|
Enzyme Inhibitors; Alimentary Tract and Metabolism; Various Alimentary Tract and Metabolism Products; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | Eliglustat is indicated for the long-term treatment of type 1 Gaucher disease in patients who are CYP2D6 extensive metabolizers (EMs), intermediate metabolizers (IMs), or poor metabolizers (PMs) in treatment-naive and treatment-experienced adult patients. |
| FDBD01652 | Cobicistat |
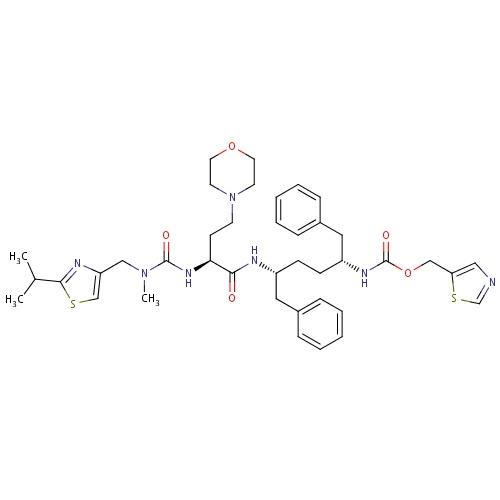
|
Anti-HIV Agents; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP3A Inhibitors; CYP2D6 Inducers; CYP2D6 Inducers (strong); | Cobicistat is a CYP3A inhibitor indicated to increase systemic exposure of atazanavir or darunavir (once daily dosing regimen) in combination with other antiretroviral agents in the treatment of HIV-1 infection. It is not interchangeable with ritonavir to increase systemic exposure of darunavir 600 mg twice daily, fosamprenavir, saquinavir, or tipranavir due to lack of exposure data. The use of cobicistat is not recommended with darunavir 600 mg twice daily, fosamprenavir, saquinavir or tipranavir. Complex or unknown mechanisms of drug interactions preclude extrapolation of ritonavir drug interactions to certain cobicistat interactions. Cobicistat and ritonavir when administered with either atazanavir or darunavir may result in different drug interactions when used with concomitant medications. |
| FDBD01692 | Zucapsaicin |
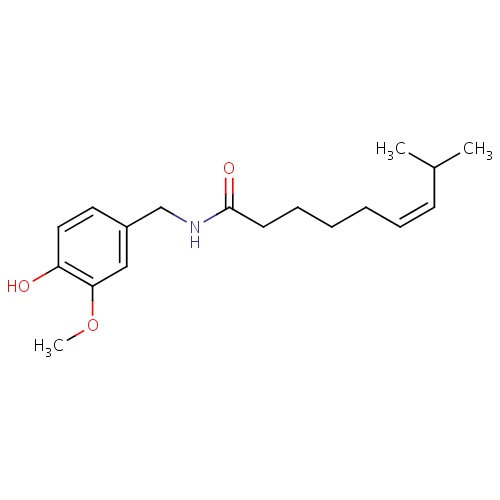
|
Musculo-Skeletal System; Topical Products for Joint and Muscular Pain; Capsaicin and Similar Agents; | |
| FDBD01702 | Iothalamic acid |
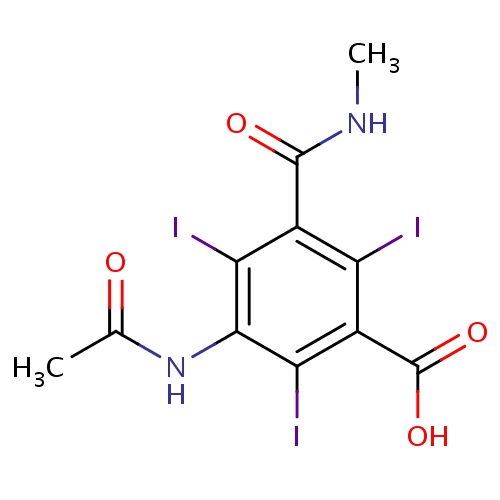
|
Contrast Media; Watersoluble, Nephrotropic, High Osmolar X-Ray Contrast Media; X-Ray Contrast Media, Iodinated; | |
| FDBD01704 | Ioxilan |
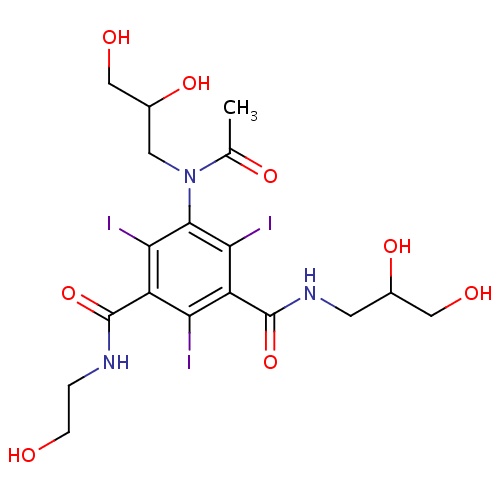
|
Contrast Media; X-Ray Contrast Media, Iodinated; Watersoluble, Nephrotropic, Low Osmolar X-Ray Contrast Media; | |
| FDBD01706 | Technetium Tc 99m Mebrofenin |
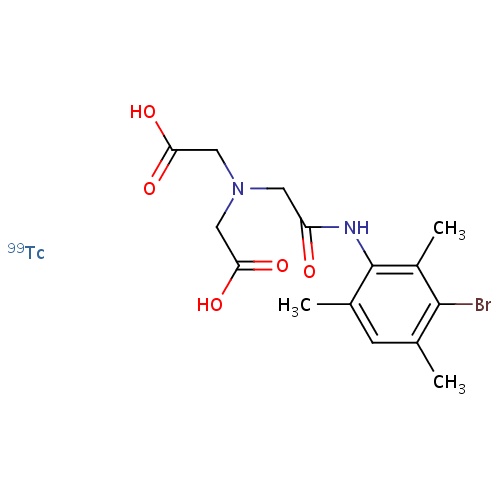
|
Diagnostic Radiopharmaceuticals; Technetium (99Mtc) Compounds; Hepatic and Reticulo Endothelial System; |
134 ,
14
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1wdn_ligand_1_2.mol2 | 1wdn | 1 | -5.99 | CC(=O)N | 4 |
| 4g4p_ligand_1_2.mol2 | 4g4p | 1 | -5.96 | C(=O)(N)C | 4 |
| 4kqp_ligand_1_2.mol2 | 4kqp | 1 | -5.96 | CC(=O)N | 4 |
| 1jak_ligand_frag_2.mol2 | 1jak | 1 | -5.94 | C(=O)(N)C | 4 |
| 2ntf_ligand_1_9.mol2 | 2ntf | 1 | -5.91 | C(=O)(N)C | 4 |
| 2jiw_ligand_frag_2.mol2 | 2jiw | 1 | -5.89 | NC(=O)C | 4 |
2564 ,
257

