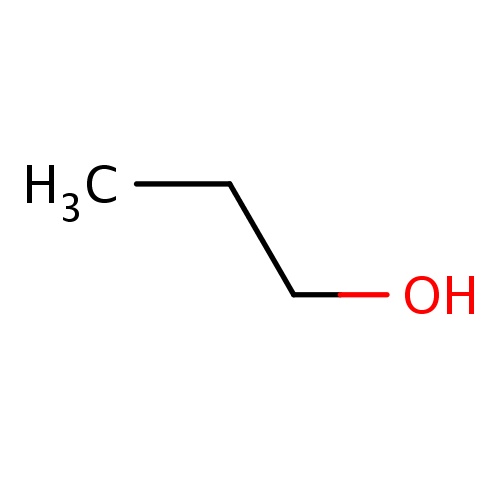
Common name
propan-1-ol
IUPAC name
propan-1-ol
SMILES
C(O)CC
Common name
propan-1-ol
IUPAC name
propan-1-ol
SMILES
C(O)CC
INCHI
InChI=1S/C3H8O/c1-2-3-4/h4H,2-3H2,1H3
FORMULA
C3H8O

Common name
propan-1-ol
IUPAC name
propan-1-ol
Molecular weight
60.095
clogP
0.038
clogS
-0.327
Frequency
0.0330
HBond Acceptor
1
HBond Donor
1
Total PolarSurface Area
20.23
Number of Rings
0
Rotatable Bond
1
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
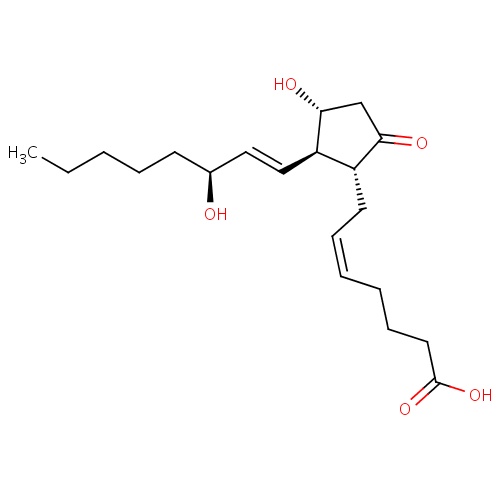
| FDBD00774 | Dinoprostone |
|
For the termination of pregnancy during the second trimester (from the 12th through the 20th gestational week as calculated from the first day of the last normal menstrual period), as well as for evacuation of the uterine contents in the management of missed abortion or intrauterine fetal death up to 28 weeks of gestational age as calculated from the first day of the last normal menstrual period. Also used in the management of nonmetastatic gestational trophoblastic disease (benign hydatidiform mole). Other indications include improving the cervical inducibility (cervical ; | |
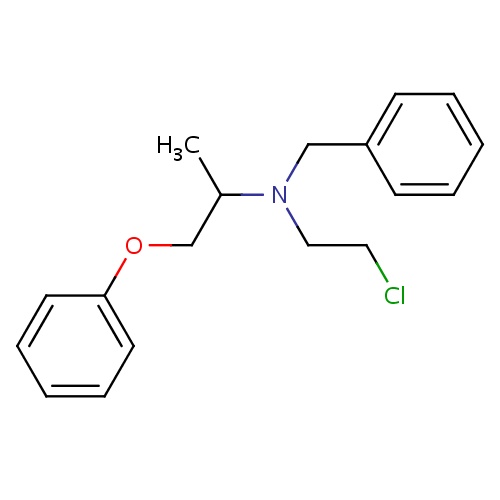
| FDBD00782 | Phenoxybenzamine |
|
Antihypertensive Agents; Vasodilator Agents; Adrenergic alpha-1 Receptor Antagonists; Adrenergic alpha-Antagonists; Cardiovascular System; Peripheral Vasodilators; CYP3A4 Inhibitors; | For the treatment of phaeochromocytoma (malignant), benign prostatic hypertrophy and malignant essential hypertension. |
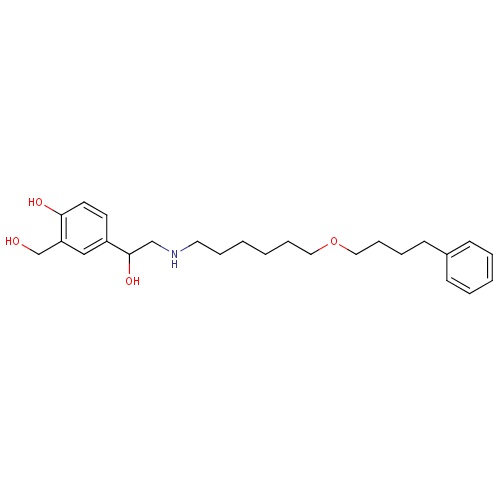
| FDBD00793 | Salmeterol |
|
Sympathomimetics; Adrenergic beta-2 Receptor Agonists; Bronchodilator Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Selective Beta-2-Adrenoreceptor Agonists; Adrenergics, Inhalants; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C8 Inducers; CYP3A4 Inhibitors; Beta2 Agonists; | For the treatment of asthma and chronic obstructive pulmonary disease (COPD). |
| FDBD00805 | Fexofenadine |
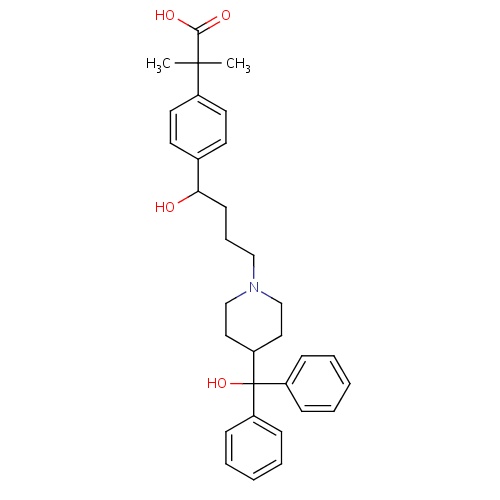
|
Anti-Allergic Agents; Histamine H1 Antagonists, Non-Sedating; Histamine Antagonists; Respiratory System; Antihistamines for Systemic Use; CYP2D6 Inducers; CYP2D6 Inducers (strong); | For management of Seasonal allergic rhinitis. |
| FDBD00827 | Ezetimibe |

|
Anticholesteremic Agents; Cholesterol Absorption Inhibitors; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; CYP3A4 Inhibitors; | For use as adjunctive therapy to diet for the reduction of elevated total-C, LDL-C, and Apo B in patients with primary (heterozygous familial and non-familial) hypercholesterolemia. |
| FDBD00862 | Busulfan |
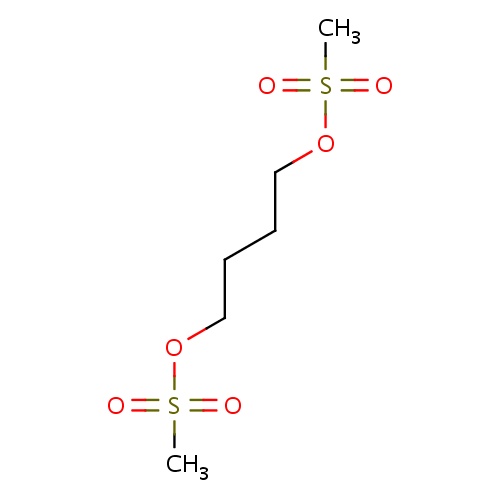
|
Antineoplastic Agents; Immunosuppressive Agents; Antineoplastic Agents, Alkylating; Alkylating Agents; Myeloablative Agonists; Antineoplastic and Immunomodulating Agents; Alkyl Sulfonates; CYP3A4 Inhibitors; | For use in combination with cyclophosphamide as a conditioning regimen prior to allogeneic hematopoietic progenitor cell transplantation for chronic myelogenous (myeloid, myelocytic, granulocytic) leukemia (FDA has designated busulfan as an orphan drug for this use). It is also used as a component of pretransplant conditioning regimens in patients undergoing bone marrow transplantation for acute myeloid leukemia and nonmalignant diseases. |
| FDBD00923 | Atazanavir |
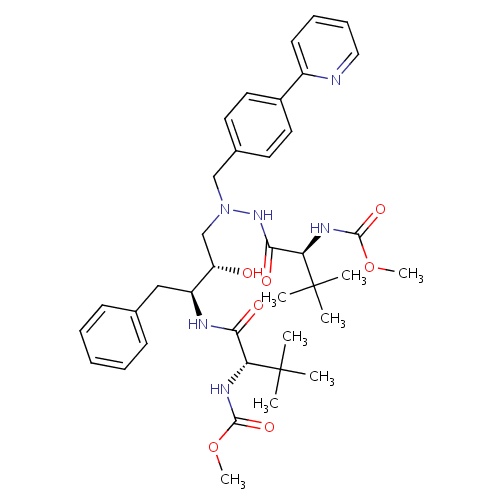
|
Anti-HIV Agents; Protease Inhibitors; HIV Protease Inhibitors; Antiinfectives for Systemic Use; Direct Acting Antivirals; Antivirals for Systemic Use; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | Used in combination with other antiretroviral agents for the treatment of HIV-1 infection, as well as postexposure prophylaxis of HIV infection in individuals who have had occupational or nonoccupational exposure to potentially infectious body fluids of a person known to be infected with HIV when that exposure represents a substantial risk for HIV transmission. |
| FDBD00927 | Atorvastatin |
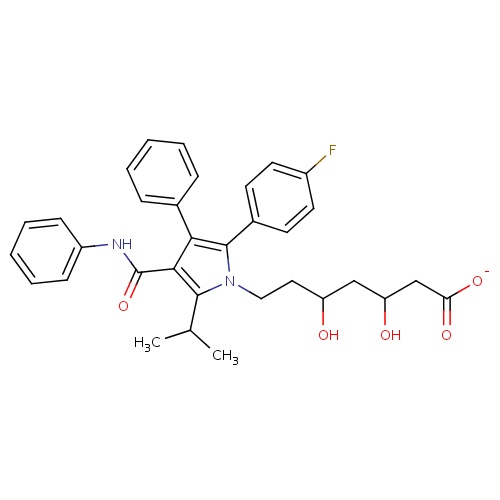
|
Anticholesteremic Agents; Hydroxymethylglutaryl-CoA Reductase Inhibitors; Dipeptidyl-Peptidase IV Inhibitors; HMG CoA Reductase Inhibitors; Lipid Modifying Agents, Plain; Lipid Modifying Agents; Cardiovascular System; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP2C8 Inhibitors; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; Cytochrome P-450 CYP2C8 Inducers; Cytochrome P-450 CYP2B6 Inducers; Cytochrome P-450 CYP2B6 Inhibitors; CYP2B6 Inhibitors (strong); CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; Combined Inhibitors of CYP3A4 and P-glycoprotein; | |
| FDBD00938 | Iloprost |
|
Platelet Aggregation Inhibitors; Vasodilator Agents; Antithrombotic Agents; Blood and Blood Forming Organs; Platelet Aggregation Inhibitors Excl. Heparin; | Used for the treatment of pulmonary arterial hypertension. |
| FDBD00978 | Rabeprazole |

|
Enzyme Inhibitors; Anti-Ulcer Agents; Proton Pump Inhibitors; Alimentary Tract and Metabolism; Musculo-Skeletal System; Drugs for Peptic Ulcer and Gastro-Oesophageal Reflux Disease (Gord); Drugs for Acid Related Disorders; Antiinflammatory and Antirheumatic Products, Non-Steroids; Antiinflammatory and Antirheumatic Products; Antiinflammatory Preparations, Non-Steroids for Topical Use; Topical Products for Joint and Muscular Pain; Butylpyrazolidines; Cytochrome P-450 CYP1A2 Inhibitors; Cytochrome P-450 CYP2C9 Inhibitors; Cytochrome P-450 CYP1A2 Inducers; Cytochrome P-450 CYP2C9 Inducers; Cytochrome P-450 CYP2C19 Inducers; CYP2D6 Inducers; CYP2D6 Inducers (strong); CYP3A4 Inhibitors; | For the treatment of acid-reflux disorders (GERD), peptic ulcer disease, H. pylori eradication, and prevention of gastroinetestinal bleeds with NSAID use. |
96 ,
10
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 3cow_ligand_1_0.mol2 | 3cow | 1 | -6.56 | C(C)(C)(C)CO | 6 |
| 4fhi_ligand_1_8.mol2 | 4fhi | 1 | -6.55 | C(O)C(C)(C)C | 6 |
| 4fhh_ligand_1_8.mol2 | 4fhh | 1 | -6.49 | C(O)C(C)(C)C | 6 |
| 2flh_ligand_1_0.mol2 | 2flh | 1 | -6.11 | C(O)C(C)C | 5 |
| 3bex_ligand_1_1.mol2 | 3bex | 1 | -6.04 | C(C)(C)CO | 5 |
| 3bex_ligand_1_0.mol2 | 3bex | 1 | -6.03 | C(C)(C)CO | 5 |
| 3guz_ligand_1_1.mol2 | 3guz | 1 | -6.03 | C(O)C(C)C | 5 |
| 4rce_ligand_2_5.mol2 | 4rce | 1 | -6.01 | OCC(C)(C)C | 6 |
967 ,
97

