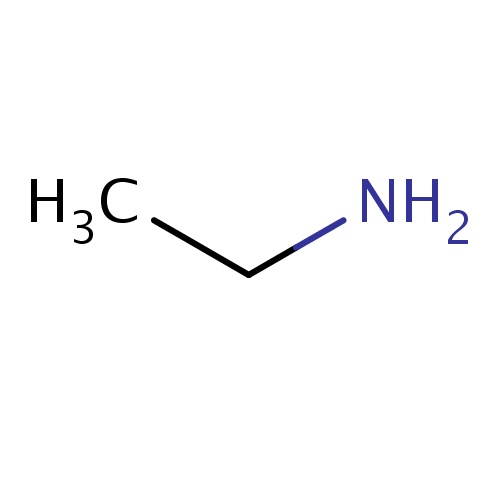
Common name
ethanamine
IUPAC name
ethanamine
SMILES
CCN
Common name
ethanamine
IUPAC name
ethanamine
SMILES
CCN
INCHI
InChI=1S/C2H7N/c1-2-3/h2-3H2,1H3
FORMULA
C2H7N

Common name
ethanamine
IUPAC name
ethanamine
Molecular weight
45.084
clogP
-0.554
clogS
-0.083
Frequency
0.0677
HBond Acceptor
0
HBond Donor
2
Total PolarSurface Area
26.02
Number of Rings
0
Rotatable Bond
0
| Drug ID | Common name | Structure CAS | Compound class | Therapeutic area |
|---|---|---|---|---|
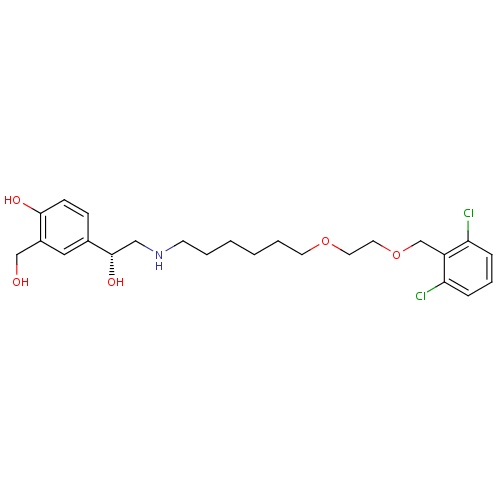
| FDBD01665 | Vilanterol |
|
Immunosuppressive Agents; Respiratory System; Drugs for Obstructive Airway Diseases; Adrenergics, Inhalants; CYP3A4 Inhibitors; Beta2 Agonists; | Vilanterol is approved for use in several combination products such as with fluticasone furoate under the tradename Breo Ellipta and in combination with umeclidinium bromide as Anoro Ellipta. Approved by the FDA in 2013, use of Breo Ellipta is indicated for the long-term, once-daily maintenance treatment of airflow obstruction in patients with COPD, including chronic bronchitis and emphysema. It is also indicated for once-daily maintenance treatment of asthma in patients aged 18 or older with reversible obstructive airways disease. |
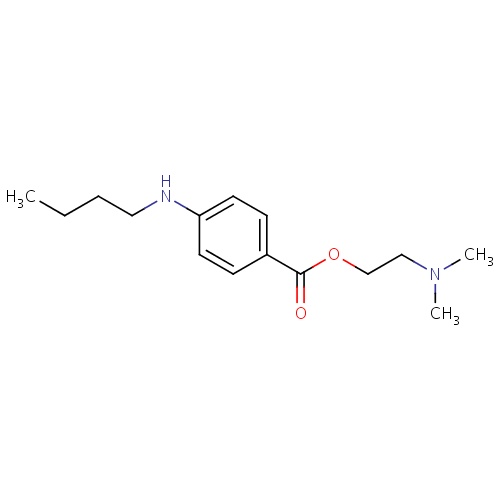
| FDBD01668 | Tetracaine |
|
Anesthetics, Local; Anesthetics; Ophthalmologicals; Sensory Organs; Dermatologicals; Nervous System; Cardiovascular System; Local Anesthetics; Agents for Treatment of Hemorrhoids and Anal Fissures for Topical Use; Vasoprotectives; Anesthetics for Topical Use; Antipruritics, Incl. Antihistamines, Anesthetics, Etc.; Esters of Aminobenzoic Acid; | |
| FDBD01679 | Quinagolide |

|
Genito Urinary System and Sex Hormones; Prolactine Inhibitors; | |
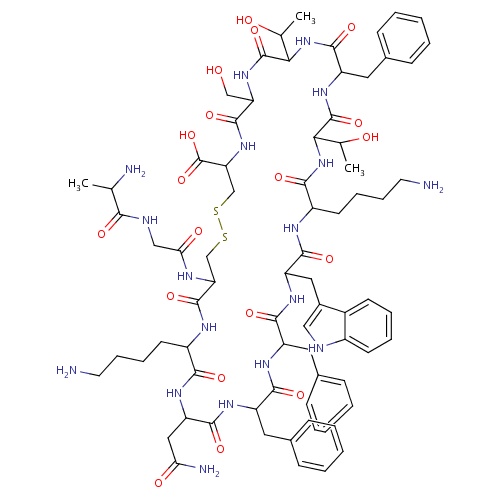
| FDBD01680 | Somatostatin |
|
Hormones; Pituitary and Hypothalamic Hormones and Analogues; Systemic Hormonal Preparations, Excl. Sex Hormones and Insulins; Hypothalamic Hormones; Somatostatin and Analogues; | For the symptomatic treatment of acute bleeding from esophageal varices. Other treatment options for long-term management of the condition may be considered if necessary, once initial control has been established. |
| FDBD01705 | Isosulfan blue |
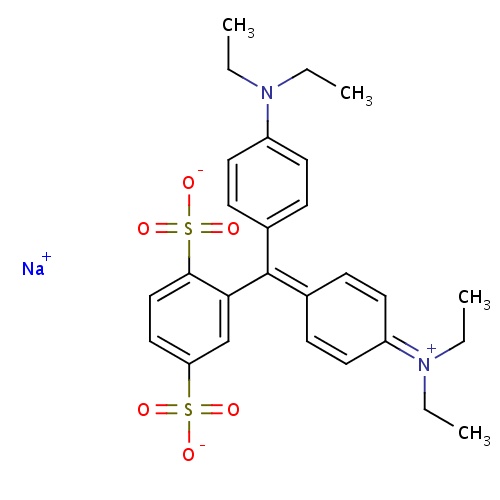
|
; | |
| FDBD01750 | Polaprezinc |
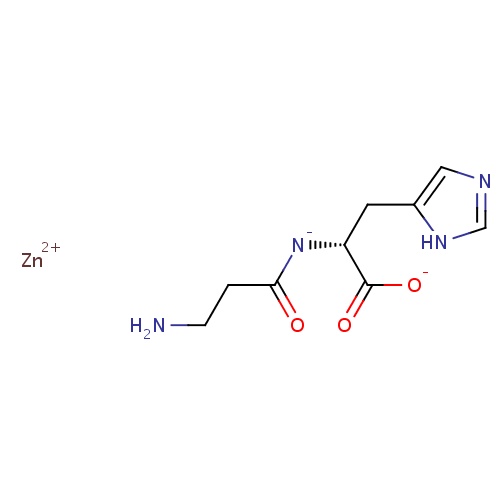
|
; | |
| FDBD01759 | Efonidipine |
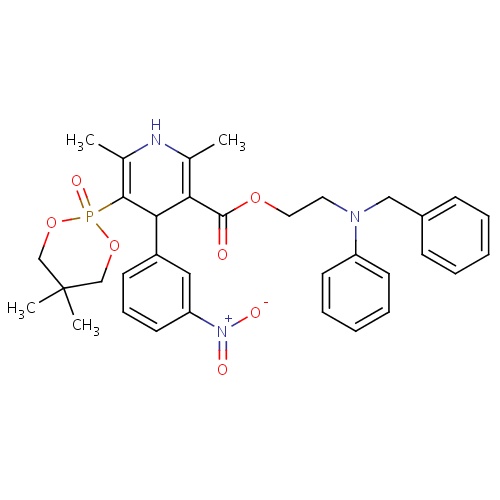
|
; | For the treatment of hypertension. |
| FDBD01761 | Levamlodipine |
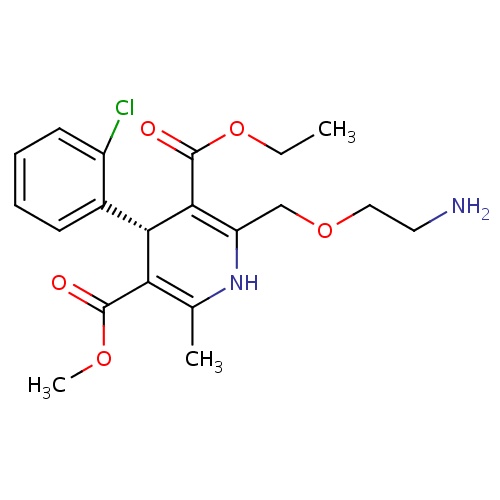
|
; | For the treatment of hypertension. |
| FDBD01769 | Lixisenatide |

|
Drugs Used in Diabetes; Alimentary Tract and Metabolism; Blood Glucose Lowering Drugs, Excl. Insulins; | For lowering blood glucose level in patients with type 2 diabetes. |
| FDBD01773 | Eluxadoline |
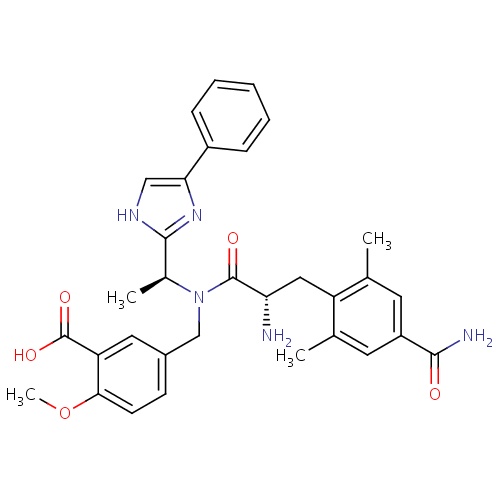
|
Gastrointestinal Agents; | For the treatment of irritable bowel syndrome with diarrhea (IBS-D). |
197 ,
20
| FRAGNAME | PDBID | SIMILIRITY | XSCORE | SMILE | HAC |
|---|---|---|---|---|---|
| 1a99_ligand_1_2.mol2 | 1a99 | 1 | -5.92 | CC[NH3+] | 3 |
| 1pot_ligand_1_5.mol2 | 1pot | 1 | -5.90 | C([NH3+])C | 3 |
| 1i7m_ligand_1_2.mol2 | 1i7m | 1 | -5.88 | CC[NH3+] | 3 |
| 3a5y_ligand_1_0.mol2 | 3a5y | 1 | -5.86 | [NH3+]CC | 3 |
| 2zxg_ligand_frag_1.mol2 | 2zxg | 1 | -5.85 | C([NH3+])C | 3 |
| 3ebh_ligand_1_0.mol2 | 3ebh | 1 | -5.84 | [NH3+]CC | 3 |
| 4q4i_ligand_1_0.mol2 | 4q4i | 1 | -5.83 | [NH3+]CC | 3 |
1149 ,
115

